|
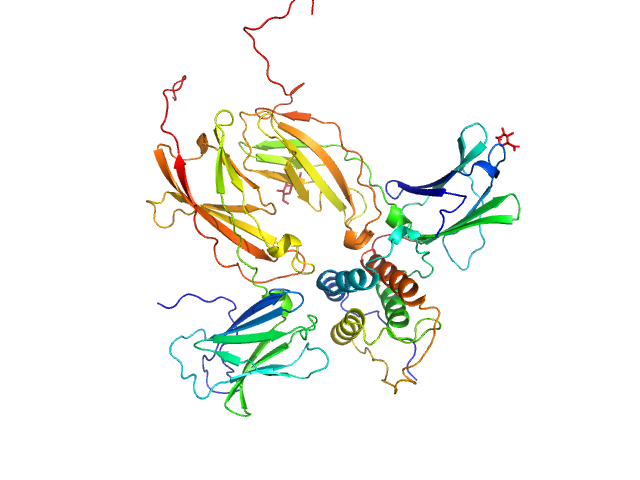
SAXS data were measured on the SWING beam line at the SOLEIL Synchrotron (Gif-sur-Yvette, France) with an online HPLC system (David and Perez, 2009). 50 µL of glycan-minimized ternary TSLPΔ127-131:TSLPRN47Q:IL-7R complex (6 mg/mL), prepared as for crystallographic studies, was injected onto an Agilent 4.6 x 300 mm Bio SEC-3 column with 300 Å pore size and HBS pH 7.4 as running buffer at a flow speed of 0.2 mL/min at 288 K. X-ray scattering data were collected in continuous flow mode with 1s exposure time per frame and an acquisition rate of 1 frame every 2 seconds. Scattering intensities, I(s), were recorded within a momentum transfer range of 0.1 nm−1 < s < 6 nm−1 (I(s) vs s, where s = 4πsinθ/λ; 2θ is the scattering angle and λ = 0.103 nm). Raw data were radially averaged and buffer subtracted using Foxtrot v3.2.7 (developed at Synchrotron Soleil and provided by Xenocs (Sassenage, France). The quality of the data was analyzed with Foxtrot by checking the stability of the radius of gyration over the length of the elution peak and by scaling all curves to the most intense scattering profile. The final scattering curve was obtained by averaging the unscaled, buffer-subtracted scattering profiles from frames 119 – 128, which correspond to the top of the elution peak. Structural parameters were determined with the ATSAS suite (Petoukhov et al., 2012). The radius of gyration was determined with autoRg v1.37. The distance distribution function was obtained by GNOM v5.0. The Porod volume was calculated with DATPOROD (Petoukhov et al., 2012). Molecular weight estimates were obtained by ScÅtter (Rambo and Tainer, 2013), SAXS-MoW2 (Fischer et al., 2010), DATPOROD and from the extrapolated scattering intensity to zero angle, I(0), on an absolute scale (using water as a reference; SWING beam line). To compare the experimental scattering profile with the theoretical scattering profile from models based on the determined X-ray structure the AllosMod-FoXS server was used (Guttman et al., 2013). 100 comparative full-atom models similar to the input structure were generated by Modeller and their scattering profile was calculated and fitted to the experimental data by FoXS to a s-max of 2.5 nm-1.
Storage temperature = UNKNOWN. Sample detector distance = UNKNOWN. Concentration min = UNKNOWN. Concentration max = UNKNOWN
|
|
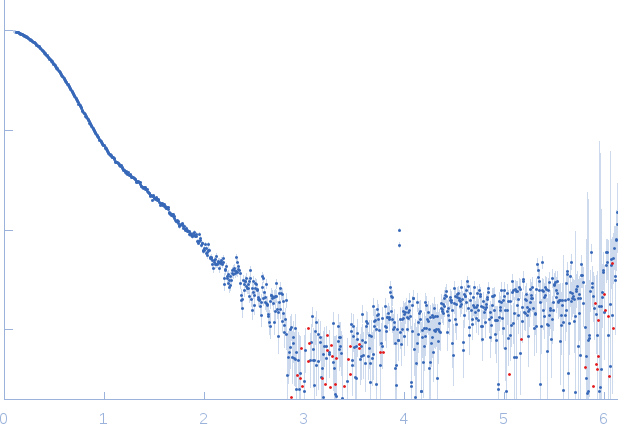 s, nm-1
s, nm-1