|
Synchrotron SAXS data from solutions of cGMP-dependent protein kinase 1 (∆53 PKG Iα) were collected using SEC-SAXS on the BL4-2 beam line at the Stanford Synchrotron Radiation Lightsource (SSRL; Stanford, CA, USA) using a Rayonix MX225-HE detector at a sample-detector distance of 1.7 m and at a wavelength of λ = 0.1127 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). A 35 μL sample of 6 mg/mL PKG Iα ∆53 was injected onto a Superdex 200 3.2/30 column (GE) and eluted isocratically using an Ettan FPLC (GE) at 0.05 mL/min in 50 mM MES, 300 mM NaCl, 1mM TCEP, pH 6.9 supplemented with 5 mM DTT as a radical scavenger. The eluant stream was connected in-line to a 1.5 mm quartz capillary. Data were collected with a 1 second/frame exposure rate at 22°C. A total of 600 frames were collected. Frames 410-459 containing the peak corresponding to ∆53 PKG were averaged and subtracted from background. The scattering curve, Guinier and P(r) functions, and Porod volume were calculated using the PRIMUS (ATSAS) program suite.
SEC column = UNKNOWN. Sample injection volume = UNKNOWN. Flow rate = UNKNOWN
|
|
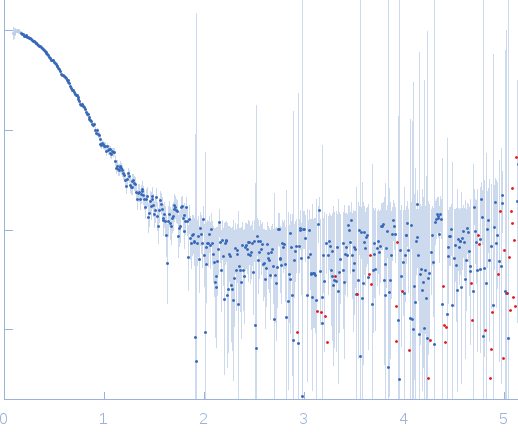 s, nm-1
s, nm-1
