|
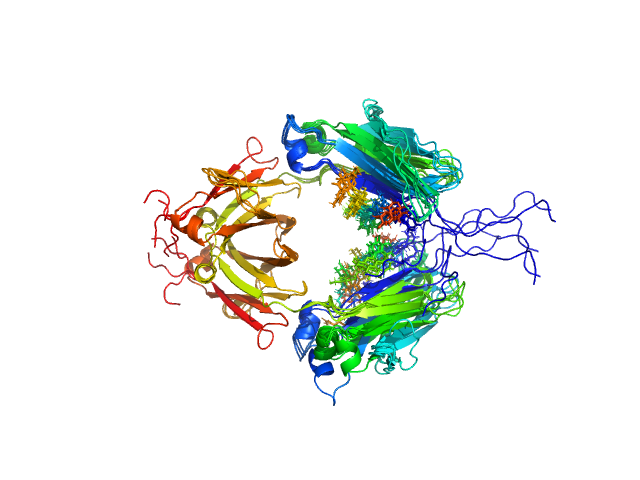
Synchrotron SAXS data from solutions of the Fc region of Immunoglobulin G1 (IgG1 Fc) in 20mM HEPES, 50mM NaCl, pH 7.5, were collected on the 12.3.1 (SIBYLS) beam line at the Advanced Light Source (ALS; Berkeley, CA, USA) using a Pilatus3 X 2M detector at a sample-detector distance of 1.5 m and at a wavelength of λ = 0.103 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Solute concentrations ranging between 1 and 14 mg/ml were measured at 20°C. 33 successive 0.300 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted. The low angle data collected at lower concentration were merged with the highest concentration high angle data to yield the final composite scattering curve.
|
|
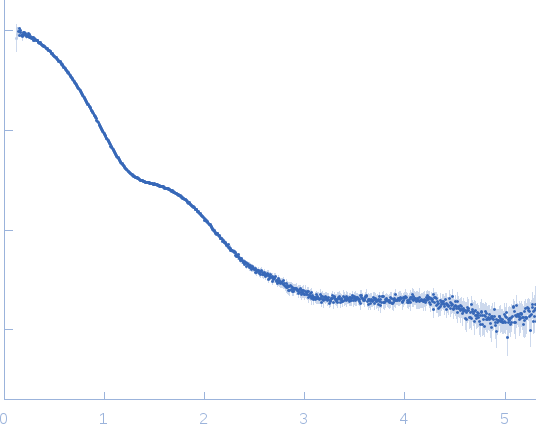 s, nm-1
s, nm-1