|
Synchrotron SAXS data from solutions of Filamin A Ig-like domains 4-6 in 20 mM Tris, 100 mM NaCl, 1 mM DTT, pH 8 were collected on the BM29 beam line at the ESRF (Grenoble, France) using a Pilatus 1M detector at a sample-detector distance of 2.9 m and at a wavelength of λ = 0.1 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Solute concentrations ranging between 2 and 4 mg/ml were measured at 20°C. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted. The low angle data collected at lower concentration were merged with the highest concentration high angle data to yield the final composite scattering curve.
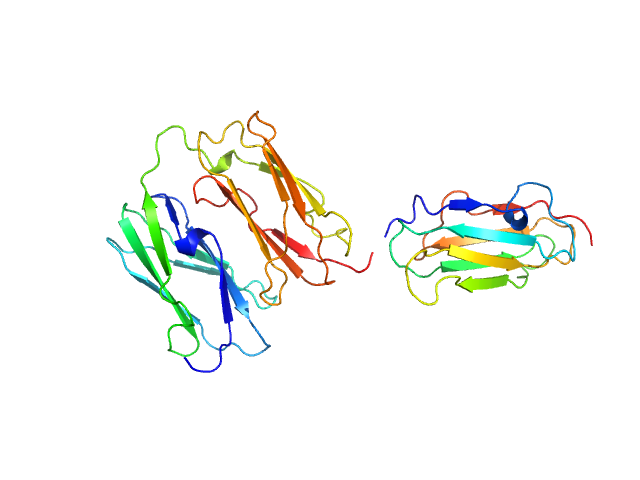
Wild type three-domain fragment of human Filamin A Ig-like domains 4-6 (FLNa4-6). There are two extra amino acids, Met and Ser, in the very N-terminus of the polypeptide that have resulted from TEV-cleavage of the GST fusion tag during the protein purification.
|
|
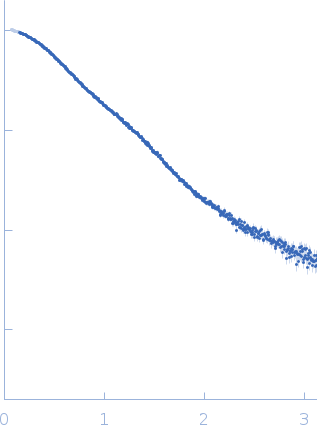 s, nm-1
s, nm-1