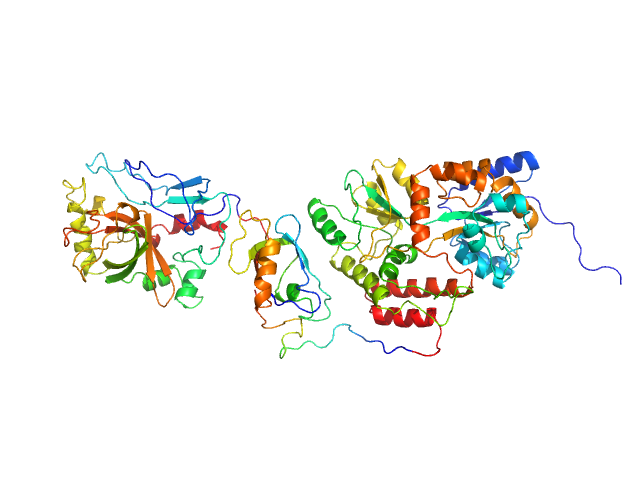
|
Synchrotron SAXS data from solutions of MBP-SpoIVB peptidase fusion protein in 20 mM Tris-HCl, 150 mM NaCl, 5% glycerol, pH 8 were collected on the BL19U2 beam line at the Shanghai Synchrotron Radiation Facility (SSRF, Shanghai, China) using a Pilatus 1M detector at a sample-detector distance of 2.3 m and at a wavelength of λ = 0.103 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). One solute concentration of 5.00 mg/ml was measured at 10°C. 20 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
The originally deposited p(r) profile is included in the full entry zip archive.
|
|
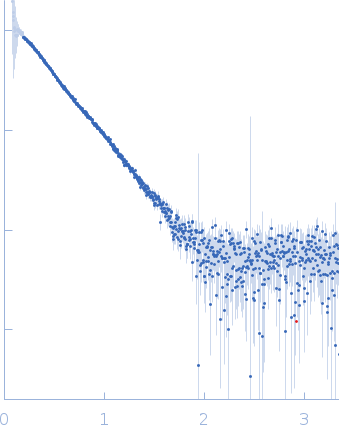 s, nm-1
s, nm-1