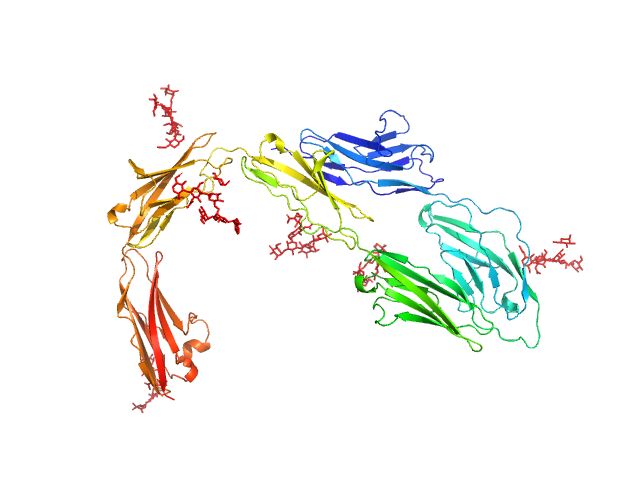
|
Synchrotron SAXS
data from solutions of
High mannose glycan contactin 1 immunoglobulin domains 1-6, 2.7 μM
in
25 mM HEPES, 150 mM NaCl, pH 7.5
were collected
on the
B21 beam line
at the Diamond Light Source storage ring
(Didcot, UK)
using a Eiger 4M detector
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
One solute concentration of 0.18 mg/ml was measured.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
High mannose glycan contactin 1 immunoglobulin domains 1-6 2.7μM. Experimental molecular weight=Sequence Expected MW + MW for 6 N glycosylation sites confirmed crystallographically. X-ray wavelength: UNKNOWN. X-ray exposure time: UNKNOWN. Experimental temperature: UNKNOWN. Dmax underestimated.
|
|
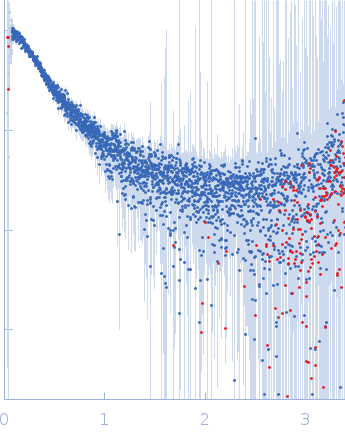 s, nm-1
s, nm-1