|
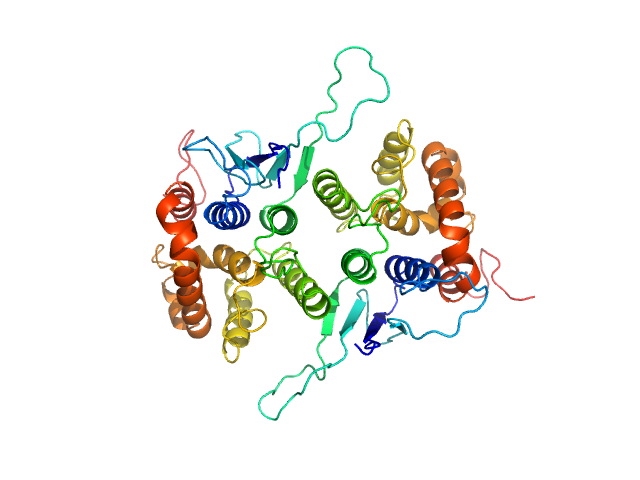
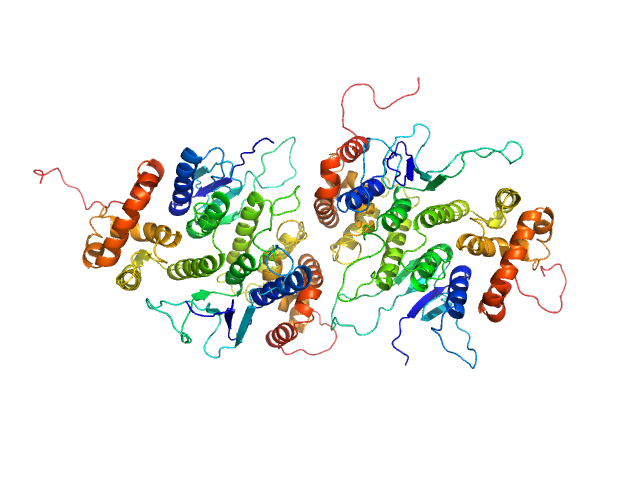
Synchrotron SAXS data of the N-terminal GST-like domain of cytosolic glutamyl-tRNA synthetase (ERS) from Plasmodium berghei. The experiment was performed on the SWING beamline at the SOLEIL synchrotron (Saint-Aubin, France) using an EigerX 4M at a sample-detector distance of 2 m and at a wavelength of λ = 0.1033 nm. In-line size-exclusion chromatography was used to separate the sample prior to X-ray exposure. 50 μl of purified recombinant (E. coli) protein at 13 mg/mL were injected on a Bio SEC-3 column (4.6 × 300 mm, 300 Å) equilibrated in 25 mM HEPES-NaOH pH 7.0, 300 mM NaCl, 5% glycerol, 0.005% (m/v) DDM, 5 mM 2-mercaptoethanol at 0.2 mL/min and 15°C. Frames (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle) were collected succesively each 1 s (0.990 s exposure and 0.01 dead time) in two time windows. 180 solvent frames were collected at time 2.5 min prior to the column's void volume and 1230 sample frames were collected from time 9.5 min until the end of the run. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent was subtracted from the sample frames using Foxtrot 3.5.10. Correction for capillary fouling was applied using the US-SOMO 4.0 HPLC SAXS module. Data analysis was performed with BioXTAS RAW 2.1.0. The experimental molecular weight was determined using the method of the volume of correlation. A correction factor has been applied to the Porod volume. The SAXS profile likely correspond to a mixture of dimers and tetramers. Models of these two species were built with MODELLER 10.1 based on the crystal structure (PDB 8BCQ) and used for OLIGOMER fitting. The volume fractions of dimer and tetramer were 0.779 and 0.221, respectively.
|
|
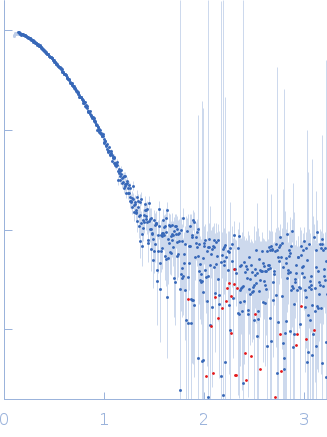 s, nm-1
s, nm-1