|
Synchrotron SAXS data from solutions of horse spleen apoferritin in 137 mM NaCl, 2.7 mM KCl, 10 mM phosphate buffer, 0.9 mM CaCl2, 0.5 mM MgCl2, pH 7.4 were collected on the BL19U2 beam line at the Shanghai Synchrotron Radiation Facility (SSRF; Shanghai, China) using a Pilatus 1M detector at a sample-detector distance of 2.7 m and at a wavelength of λ = 0.1033 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 80.00 μl sample at 2.1 mg/ml was injected at a 0.50 ml/min flow rate onto a GE Superose 6 Increase 10/300 column at 20°C. 40 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
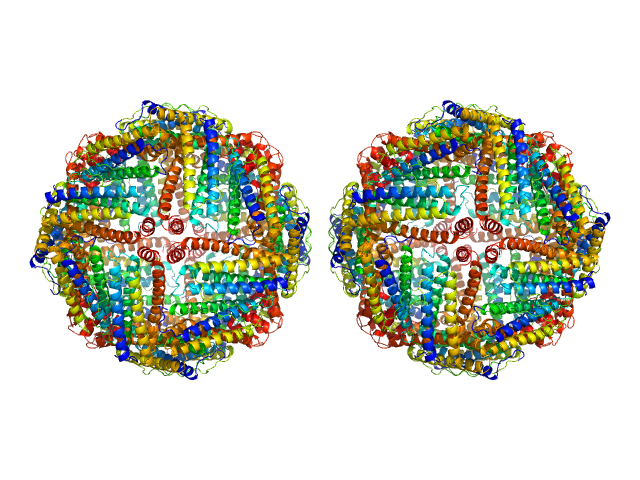
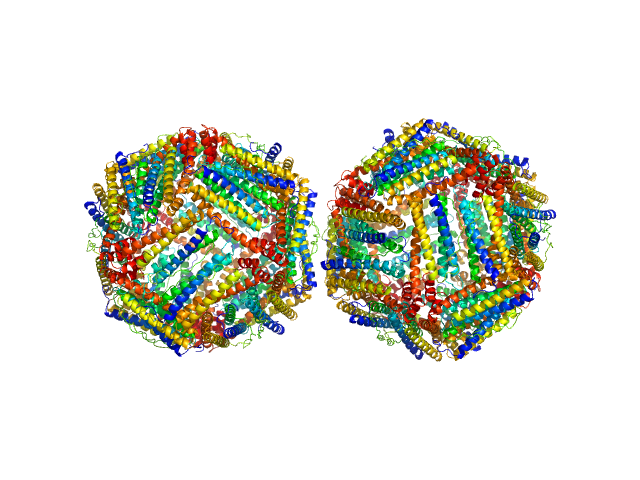
The preparation and analysis of horse spleen apoferritin were conducted to isolate its monomeric (24mer) and dimeric (2 x 24mer) forms and enable detailed structural characterization.
Sample Preparation:
Horse spleen apoferritin (purchased from Sigma-Aldrich, Merck KGaA, Germany), supplied with 50% v/v glycerol, was dialyzed overnight against 200 mM sodium acetate (pH 5.0). The dialyzed sample was then heated at 50°C for 5 hours, followed by centrifugation at 20,000 rcf for 15 minutes. The resulting supernatant was injected into a Superose 6 column (24 ml).
Fractionation and Concentration:
Fractions corresponding to apoferritin 24mer dimers were pooled and neutralized by adding an appropriate volume of 1M Tris (pH 8.0). The sample was concentrated to a final concentration of 2.1 mg/ml using a 100 kDa centrifuge spin filter and centrifuged again at 10,000 rcf for 10 minutes.
Size Exclusion Chromatography and SAXS Analysis:
Approximately 80 μl of the supernatant were loaded onto a Superose 6 Increase 10/300 column (GE Healthcare), equilibrated in DPBS buffer (137 mM NaCl, 2.7 mM KCl, 10 mM phosphate buffer, pH 7.4, 0.9 mM CaCl2, 0.5 mM MgCl2). The flow rate was set at 0.5 ml/min. SAXS frames corresponding to the dimeric (2 x 24mer) fraction of apoferritin (elution volume range: 12.9–13.25 ml) with a constant Rg value were selected for further analysis. These dimers are assumed to be predominantly disulfide-bonded, as dynamic dimers tend to dissociate into 24mer monomers, reaching a dynamic equilibrium (the contribution of dynamic 2 x 24mer dimers is estimated <15% of the overall dimers).
|
|
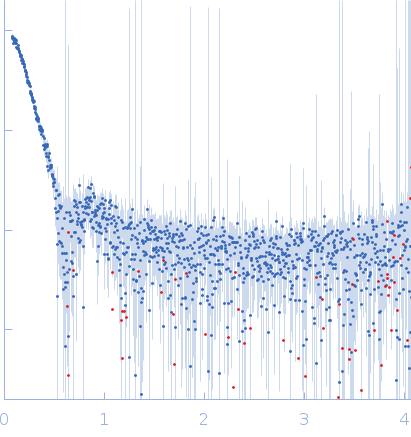 s, nm-1
s, nm-1