|
Synchrotron SAXS
data from solutions of
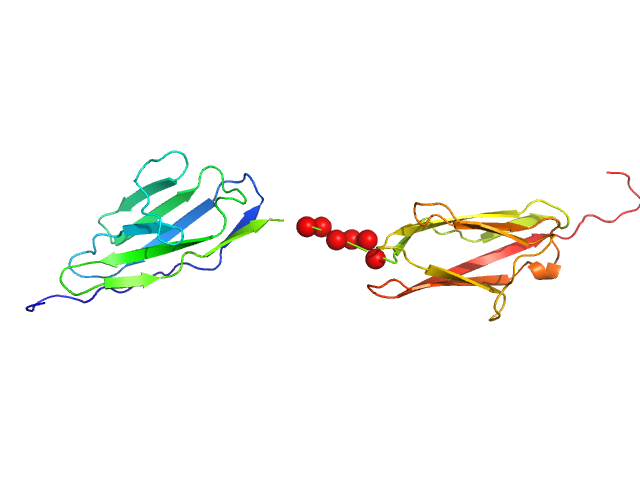
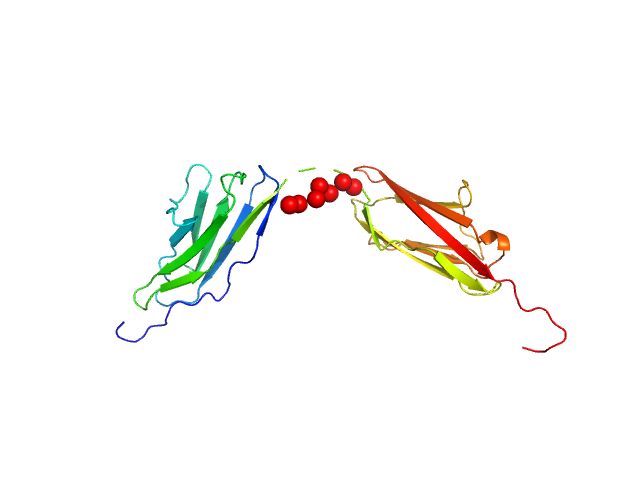
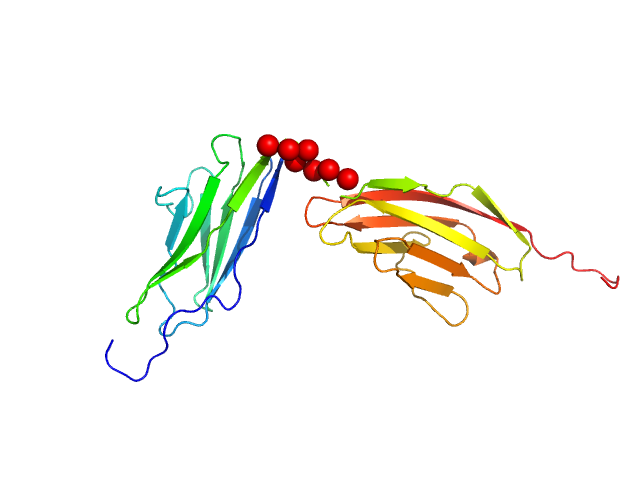
Palladin Ig3-Ig4 tandem domain with linker swapped for the shorter Ig4-Ig5 Linnker
in
HEPES, pH 7.4
were collected
on the
BL4-2 beam line
at the Stanford Synchrotron Radiation Lightsource (SSRL) storage ring
(Menlo Park, CA, USA)
using a Rayonix MX225-HE detector
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
The low angle data collected at lower concentration were merged with the highest concentration high angle data to yield the final composite scattering curve.
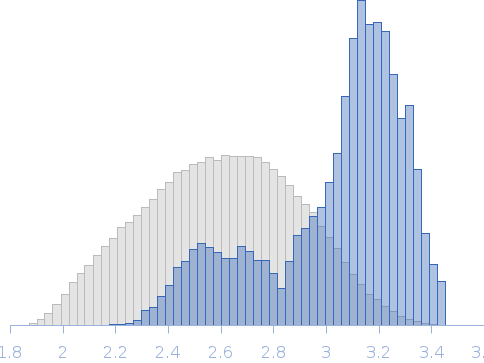
Synchrotron SAXS data from solutions of the Ig-like C2-type 3 and Ig-like C2-type 4 domains (Ig34) of palladin where the wildtype linker region has been replaced by shorter linker found between Ig4 and Ig5 domains in 20 mM HEPES pH 7.4, 1 mM DTT, 100 mM NaCl were collected on the BL4-2 beam line at the Stanford Synchrotron Radiation Lightsource (SSR; Menlo Park, CA, USA) using a Rayonix MX225-HE detector at a sample-detector distance of 1.2 m and at a wavelength of λ = 0.113 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Solute concentrations ranging between 0.5 and 10.0 mg/ml were measured at 20°C. 12 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted. The low angle data collected at lower concentration were merged with the highest concentration high angle data to yield the final composite scattering curve.
|
|
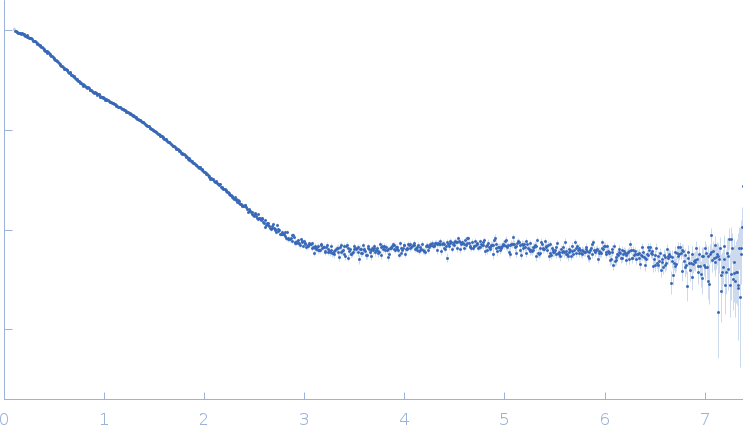 s, nm-1
s, nm-1
 Rg, nm
Rg, nm