| MWexperimental | 7660 | kDa |
| MWexpected | 7747 | kDa |
| VPorod | 16650 | nm3 |
|
log I(s)
3.95×102
3.95×101
3.95×100
3.95×10-1
|
 s, nm-1
s, nm-1
|
|
|
|
![Static model image Major vault protein OTHER [STATIC IMAGE] model](/media//pdb_file/SASDXJ3_fit1_model1.png)
|
|
![Static model image Major vault protein OTHER [STATIC IMAGE] model](/media//pdb_file/SASDXJ3_fit2_model1.png)
|
|
![Static model image Major vault protein OTHER [STATIC IMAGE] model](/media//pdb_file/SASDXJ3_fit3_model1.png)
|
|
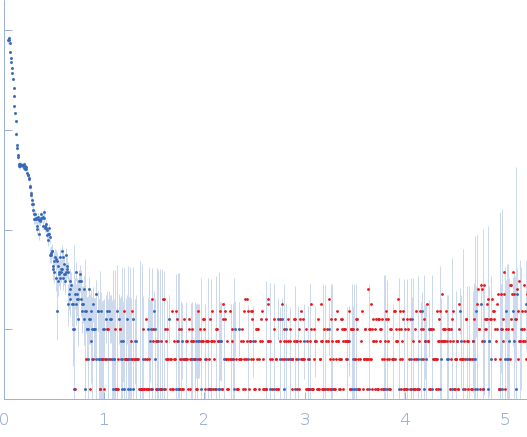
Synchrotron SAXS data from solutions of human major vault protein in 25 mM HEPES, 150 mM NaCl, 1 mM TCEP, pH 7.5 were collected on the BM29 beam line at the ESRF (Grenoble, France) using a Pilatus3 2M detector at a sample-detector distance of 2.9 m and at a wavelength of λ = 0.0992 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Two samples at a concentration of 0.5 mg/mL were measured at 20°C. For each sample measurement, ten X-ray scattering measurements with 1-second exposure times were collected. The data were normalized to the intensity of the transmitted beam and radially averaged. Ten analogous sample frames were averaged, and buffer scattering was subtracted from the sample data.
All samples were centrifuged at 20,000 × g for 10 min at 4°C prior analysis. Guinier analysis cannot be reliable performed due to the size of the particle. The atomic coordinate files of the models displayed in this entry are made available in the 'Additional files' folder of the full entry zip archive. |
|
|||||||||||||||||||||||||||