|
Synchrotron SAXS
data from solutions of
Kinase domain of Aurora kinase A mutant C290A:C393A
in
20mM HEPES, 150mM NaCl, 5mM MgCl2, 3% v/v glycerol, 2mM TCEP, pH 7.5
were collected
on the
EMBL P12 beam line
at the PETRA III storage ring
(DESY; Hamburg, Germany)
using a Pilatus 6M detector
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
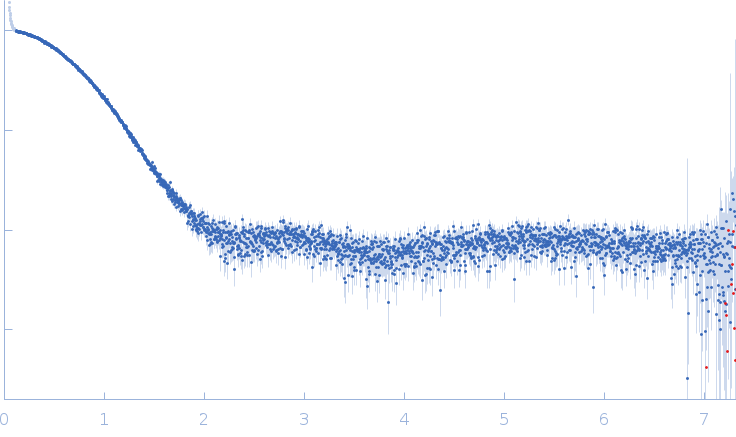
Synchrotron SAXS data from solutions of the kinase domain of Aurora kinase A (residues 122-403, containing a C290A:C393A double mutation) was collected in 20mM HEPES, 150mM NaCl, 5mM MgCl2, 3% v/v glycerol, 2mM TCEP, pH 7.5 on the EMBL P12 beam line at PETRA III (DESY, Hamburg, Germany) using a Pilatus 6M detector at a sample-detector distance of 3 m and a wavelength of λ = 0.124 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAXS was employed by injecting 80.00 μl sample of 14 mg/ml onto a GE Superdex 75 Increase 10/300 column with a flow rate of 0.70 ml/min at 20°C. 2100 successive 1 second frames were collected. The data was normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted. Buffer subtraction was done using CHROMIXS and data analysis was done using the ATSAS suite. The experimental molecular weight was also determined from SEC-MALLS run in parallel to SEC-SAXS.
|
|
 s, nm-1
s, nm-1