|
Synchrotron SAXS data from solutions of Phospholipase A and acyltransferase 3 at neutral pH in 20 mM HEPES,150 mM NaCl, 2 mM TCEP, 1% v/v glycerol, pH 7.4 were collected on the EMBL P12 beam line at PETRA III (DESY; Hamburg, Germany) using a Pilatus 6M detector at a sample-detector distance of 4 m and at a wavelength of λ = 0.1549 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 40.00 μl sample at 11.6 mg/ml was injected at a 0.35 ml/min flow rate onto a GE Superdex 75 Increase 5/150 column at 20°C. 2880 successive 0.240 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
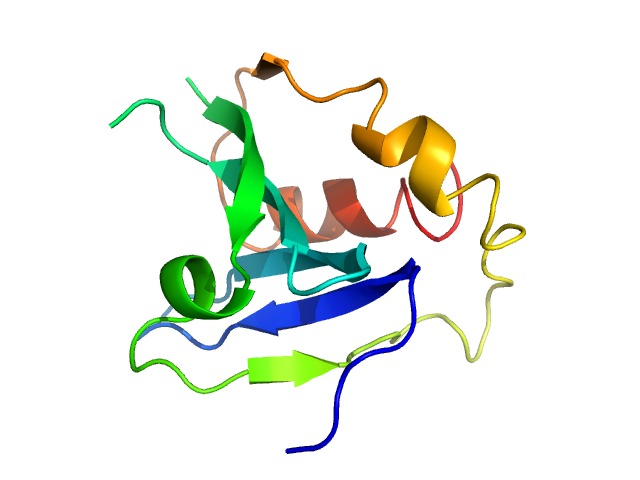
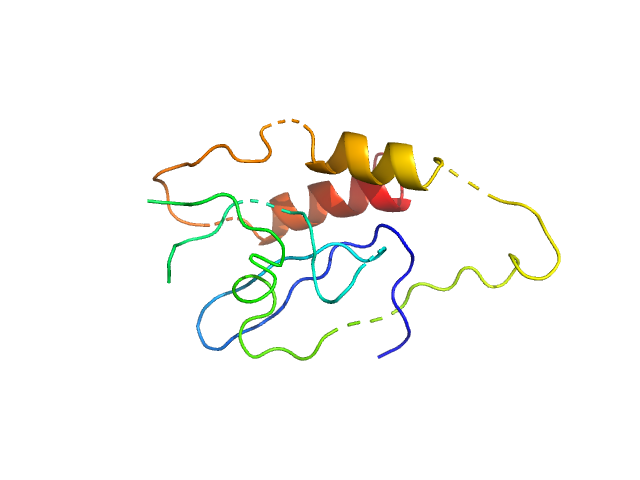
Data were processed using the ATSAS suite. The high resolution structure of PLAAT3 NTD theoretical scattering curve was fitted to the experimental data using rigid body and normal mode analysis (SREFLEX). CAUTION: The monomer component of the models (12.1 kDa) does not include all of the amino acids of the protein construct used for SAXS (14.8 kDa). CAUTION: Severe distortion of peptide geometry is noted in the SREFLEX model.
|
|
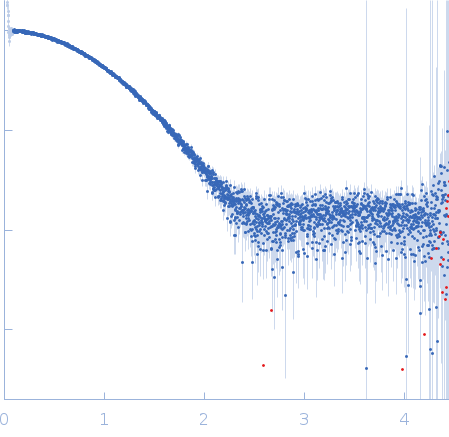 s, nm-1
s, nm-1