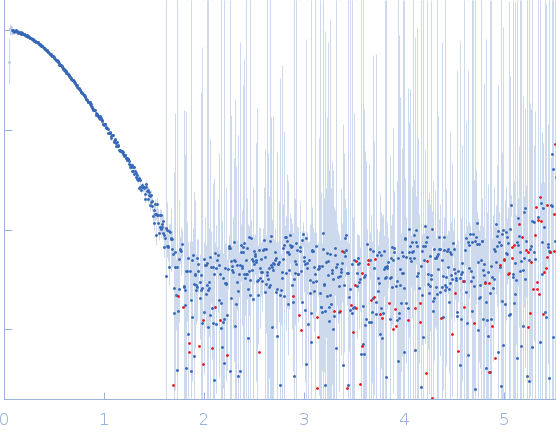
| MWexperimental | 60 | kDa |
| MWexpected | 59 | kDa |
| VPorod | 99 | nm3 |
|
log I(s)
1.32×10-2
1.32×10-3
1.32×10-4
1.32×10-5
|
 s, nm-1
s, nm-1
|
|
|
|
|
|
|
|

|
|
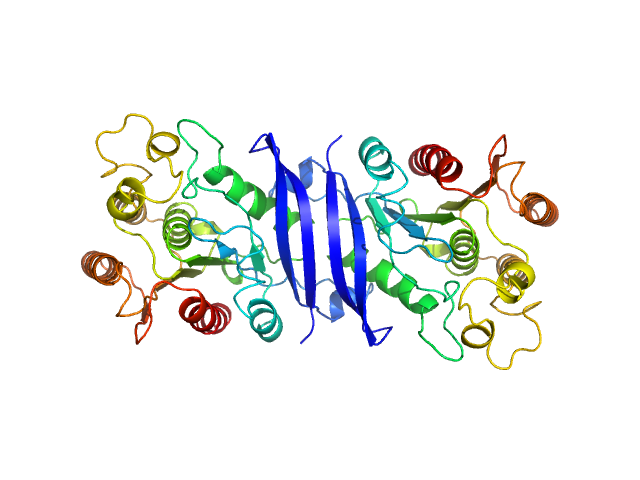
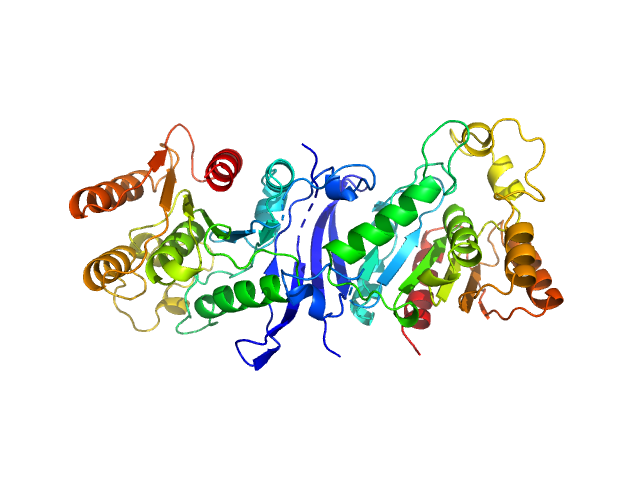
Synchrotron SAXS data from solutions of carboxylic ester hydrolase FphF in 10 mM HEPES, 50 mM NaCl, 0.1% w/v NaN3, pH 7.5 were collected on the BioSAXS beam line at the Australian Synchrotron (Melbourne, Australia) using a Pilatus3 X 2M detector at a sample-detector distance of 2.5 m and at a wavelength of λ = 0.1 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Co-flow in-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 50.00 μl sample at 6.5 mg/ml was injected at a 0.40 ml/min flow rate onto a GE Superdex 75 Increase 5/150 column at 21°C. 641 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
|
|
|||||||||||||||||||||||||||||||||||||||