|
|
|
|

|
| Sample: |
full length CtBP3 tetramer, 170 kDa protein
|
| Buffer: |
25 mM Tris/HCl 250 mM NaCl, pH: 8
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2004 Nov 18
|
The C-terminal domain of the transcriptional corepressor CtBP is intrinsically unstructured.
Protein Sci 15(5):1042-50 (2006)
Nardini M, Svergun D, Konarev PV, Spanò S, Fasano M, Bracco C, Pesce A, Donadini A, Cericola C, Secundo F, Luini A, Corda D, Bolognesi M
|
| RgGuinier |
5.1 |
nm |
| Dmax |
19.0 |
nm |
| VolumePorod |
330 |
nm3 |
|
|
|
|
|
|

|
| Sample: |
C-term part CtBP3 dimer, 52 kDa protein
|
| Buffer: |
25 mM Tris/HCl 250 mM NaCl, pH: 8
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2004 Nov 18
|
The C-terminal domain of the transcriptional corepressor CtBP is intrinsically unstructured.
Protein Sci 15(5):1042-50 (2006)
Nardini M, Svergun D, Konarev PV, Spanò S, Fasano M, Bracco C, Pesce A, Donadini A, Cericola C, Secundo F, Luini A, Corda D, Bolognesi M
|
| RgGuinier |
5.5 |
nm |
| Dmax |
20.0 |
nm |
| VolumePorod |
126 |
nm3 |
|
|
|
|
|
|

|
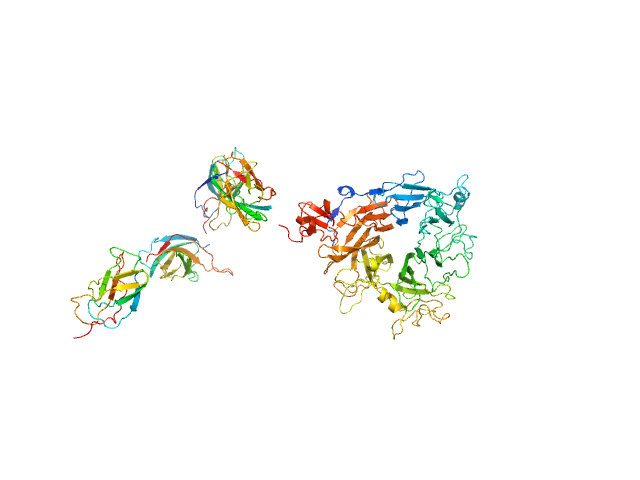
| Sample: |
Hepatocyte growth factor monomer, 79 kDa Mus musculus protein
Hepatocyte growth factor receptor monomer, 100 kDa Mus musculus protein
|
| Buffer: |
50 mM MES, 150 mM NaCl, pH: 6.7
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2004 Feb 9
|
Structural basis of hepatocyte growth factor/scatter factor and MET signalling
Proceedings of the National Academy of Sciences 103(11):4046-4051 (2006)
Gherardi E, Sandin S, Petoukhov M, Finch J, Youles M, Ofverstedt L, Miguel R, Blundell T, Vande Woude G, Skoglund U, Svergun D
|
| RgGuinier |
6.7 |
nm |
| Dmax |
20.0 |
nm |
| VolumePorod |
370 |
nm3 |
|
|
|
|
|
|
|
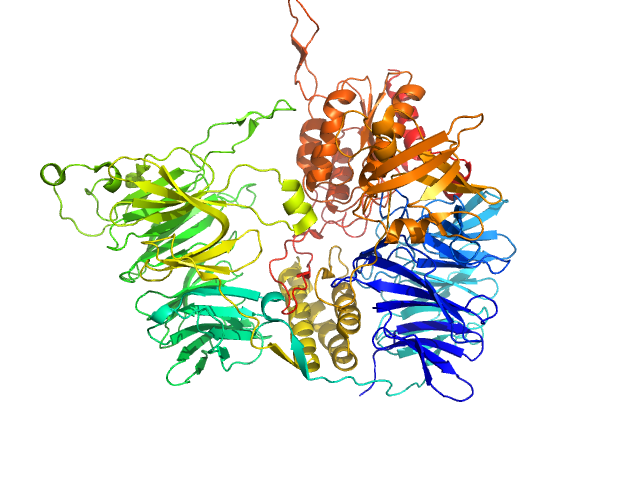
| Sample: |
Hepatocyte growth factor receptor monomer, 100 kDa Mus musculus protein
|
| Buffer: |
50 mM MES, 150 mM NaCl, pH: 6.7
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2004 Feb 7
|
Structural basis of hepatocyte growth factor/scatter factor and MET signalling
Proceedings of the National Academy of Sciences 103(11):4046-4051 (2006)
Gherardi E, Sandin S, Petoukhov M, Finch J, Youles M, Ofverstedt L, Miguel R, Blundell T, Vande Woude G, Skoglund U, Svergun D
|
| RgGuinier |
4.8 |
nm |
| Dmax |
16.0 |
nm |
| VolumePorod |
190 |
nm3 |
|
|
|
|
|
|
|
| Sample: |
Tricorn protease hexamer, 730 kDa Thermoplasma acidophilum (strain … protein
|
| Buffer: |
20mM Tris-HCl, 100 mM NaCl, pH: 7.5
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2003 Jan 14
|
X-ray snapshots of peptide processing in mutants of tricorn-interacting factor F1 from Thermoplasma acidophilum.
J Biol Chem 280(39):33387-96 (2005)
Goettig P, Brandstetter H, Groll M, Göhring W, Konarev PV, Svergun DI, Huber R, Kim JS
|
|
|
|
|
|
|

|
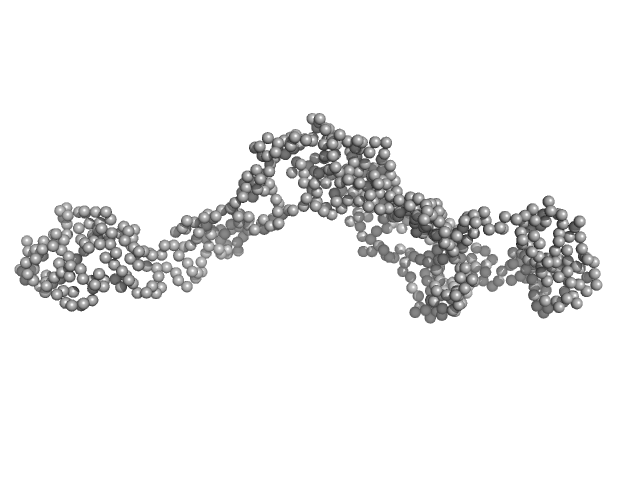
| Sample: |
Polypyrimidine tract-binding protein 2 monomer, 57 kDa Homo sapiens protein
|
| Buffer: |
25 mM Tris, 250 mM NaCl, 2 mM DTT, pH: 7.2
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2004 Feb 11
|
Structure and RNA interactions of the N-terminal RRM domains of PTB.
Structure 12(9):1631-43 (2004)
Simpson PJ, Monie TP, Szendröi A, Davydova N, Tyzack JK, Conte MR, Read CM, Cary PD, Svergun DI, Konarev PV, Curry S, Matthews S
|
| RgGuinier |
4.0 |
nm |
| Dmax |
14.0 |
nm |
| VolumePorod |
117 |
nm3 |
|
|
|
|
|
|
|
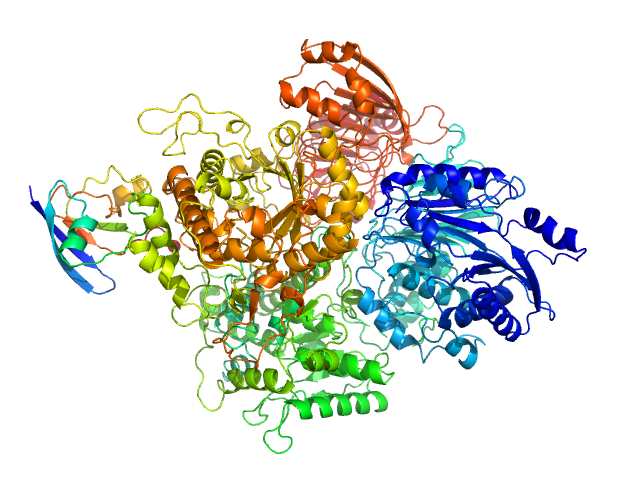
| Sample: |
Tyrosine-protein kinase BTK (R28C mutant) monomer, 76 kDa Homo sapiens protein
|
| Buffer: |
20 mM HEPES 200 mM NaCl, 2 mM DTT and 1 mM MgCl2, pH: 7.5
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2002 Apr 2
|
Conformation of full-length Bruton tyrosine kinase (Btk) from synchrotron X-ray solution scattering.
EMBO J 22(18):4616-24 (2003)
Márquez JA, Smith CI, Petoukhov MV, Lo Surdo P, Mattsson PT, Knekt M, Westlund A, Scheffzek K, Saraste M, Svergun DI
|
| RgGuinier |
5.0 |
nm |
| Dmax |
20.0 |
nm |
| VolumePorod |
130 |
nm3 |
|
|
|
|
|
|
|
| Sample: |
Ferredoxin-dependent glutamate synthase 2 monomer, 169 kDa Synechocystis sp. (strain … protein
Ferredoxin-1 monomer, 11 kDa Nostoc sp. (strain … protein
|
| Buffer: |
Hepes– KOH buffer, pH: 7.5
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2002 Jun 4
|
The Active Conformation of Glutamate Synthase and its Binding to Ferredoxin
Journal of Molecular Biology 330(1):113-128 (2003)
van den Heuvel R, Svergun D, Petoukhov M, Coda A, Curti B, Ravasio S, Vanoni M, Mattevi A
|
| RgGuinier |
3.6 |
nm |
| Dmax |
12.3 |
nm |
| VolumePorod |
262 |
nm3 |
|
|
|
|
|
|

|
| Sample: |
Procollagen C-endopeptidase enhancer 1 monomer, 46 kDa Homo sapiens protein
|
| Buffer: |
20 mM Hepes, 500 mM NaCl, pH: 7.4
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 2002 Jul 9
|
Low Resolution Structure Determination Shows Procollagen C-Proteinase Enhancer to be an Elongated Multidomain Glycoprotein
Journal of Biological Chemistry 278(9):7199-7205 (2003)
Bernocco S, Steiglitz B, Svergun D, Petoukhov M, Ruggiero F, Ricard-Blum S, Ebel C, Geourjon C, Deléage G, Font B, Eichenberger D, Greenspan D, Hulmes D
|
| RgGuinier |
4.1 |
nm |
| Dmax |
15.0 |
nm |
| VolumePorod |
117 |
nm3 |
|
|
|
|
|
|
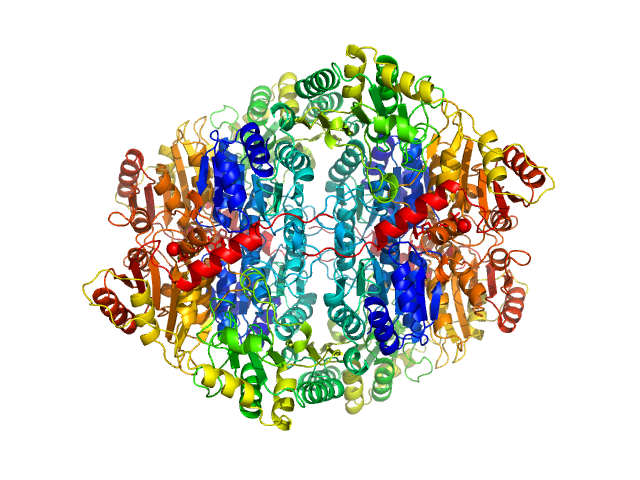
|
| Sample: |
Pyruvate decarboxylase tetramer, 244 kDa Zymomonas mobilis protein
|
| Buffer: |
100 mM Sodium Citrate, 17% Glycerol, 22.5% PEG 1500, pH: 6
|
| Experiment: |
SAXS
data collected at EMBL X33, DORIS III, DESY on 1998 Nov 3
|
Crystal versus solution structures of thiamine diphosphate-dependent enzymes.
J Biol Chem 275(1):297-302 (2000)
Svergun DI, Petoukhov MV, Koch MH, König S
|
| RgGuinier |
3.9 |
nm |
| Dmax |
11.0 |
nm |
|
|