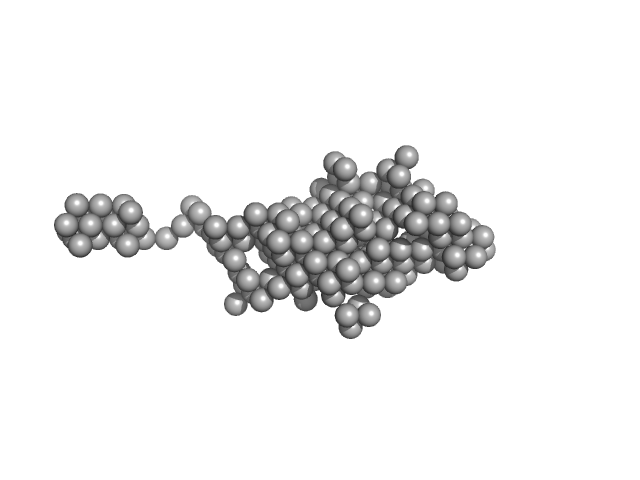
|
Synchrotron SAXS
data from solutions of
Human NEI like DNA glycosylase 1 (NEIL1) bound to DNA
in
25mM HEPES 100mM NaCl 1mM DTT, pH 7.5
were collected
on the
12.3.1 (SIBYLS) beam line
at the Advanced Light Source (ALS) storage ring
(Berkeley, CA, USA)
using a Pilatus3 X 2M detector
at a sample-detector distance of 1.4 m and
at a wavelength of λ = 1.127 nm
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
Solute concentrations ranging between 1.3 and 2.1 mg/ml were measured
at 10°C.
24 successive
0.200 second frames were collected.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
The theoretical MW for this NEIL1/DNA complex is 55.8kDa.
|
|
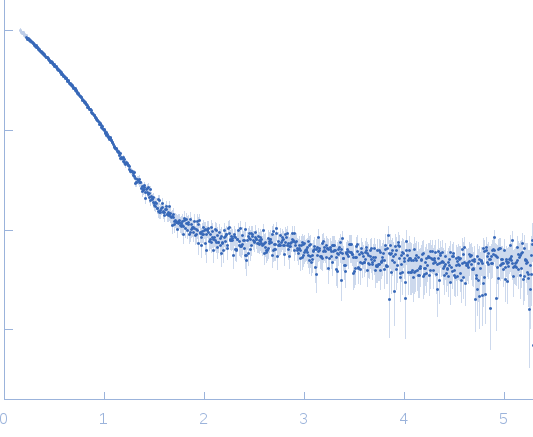 s, nm-1
s, nm-1