|
Synchrotron SAXS data from solutions of the citrate-binding PAS domain from the sensor histidine kinase, CitA, fused to lipase EstA in 10 mM glycine buffer, 10 mM NaCl, 1 mM sodium citrate, pH 10 were collected on the BM29 beam line at the ESRF (Grenoble, France) using a Pilatus 1M detector at a sample-detector distance of 2.8 m and at a wavelength of λ = 0.09919 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). One solute concentration of 5.01 mg/ml was measured at 20°C. 10 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
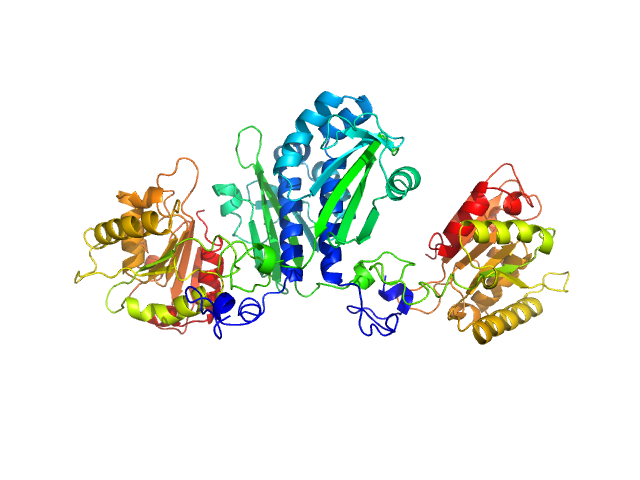
The protein is a fusion of: i) CitAP - the PAS domain from Sensor histidine kinase, CitA, from Klebsiella pneumoniae; UniProt: P52687 (Amino acid range 45-177); ii) BsLA - the Lipase EstA from Bacillus subtilis 168; UniProt: I6V559 (Amino acid range 32-212) and; iii) A linker connecting CitAP with EstA - derived from the blue light photoreceptor, YtvA, from Bacillus subtilis 168; UniProt: O34627 (Amino acid range 132-147).
|
|
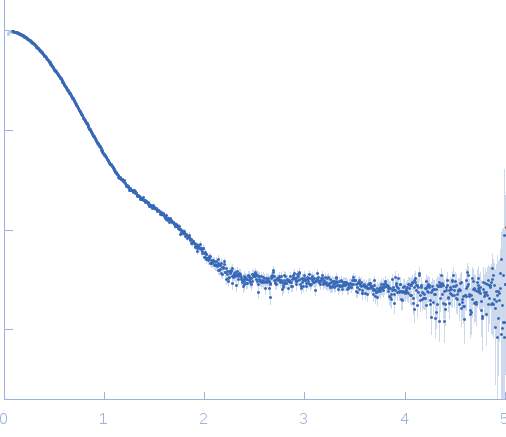 s, nm-1
s, nm-1