| MWI(0) | 17 | kDa |
| MWexpected | 16 | kDa |
| VPorod | 16 | nm3 |
|
log I(s)
6.29×102
6.29×101
6.29×100
6.29×10-1
|
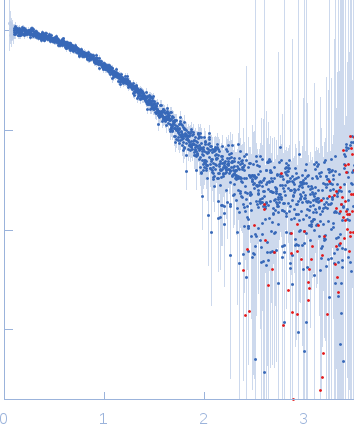 s, nm-1
s, nm-1
|
|
|
|
|
|
Synchrotron SAXS
data from solutions of
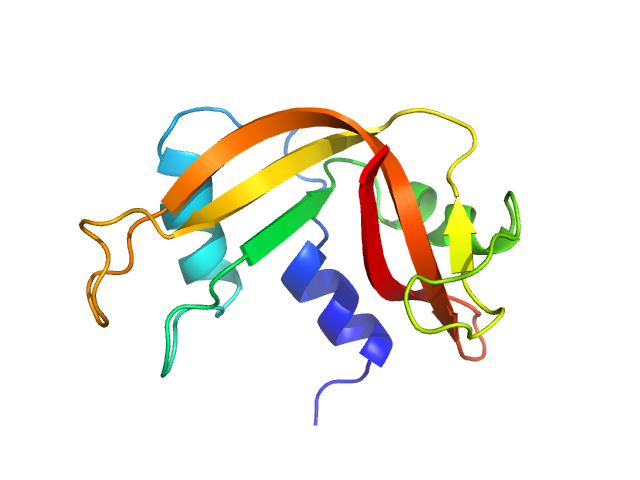
Folded ribonuclease A (RNAse)
in
phosphate buffered saline (PBS), pH 7
were collected
on the
EMBL P12 beam line
at the PETRA III storage ring
(DESY; Hamburg, Germany)
using a Pilatus 2M detector
at a sample-detector distance of 3.1 m and
at a wavelength of λ = 0.124 nm
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
One solute concentration of 5.24 mg/ml was measured
at 10°C.
Two successive
0.050 second frames were collected.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
|
|
|||||||||||||||||||||||||||||||||