|
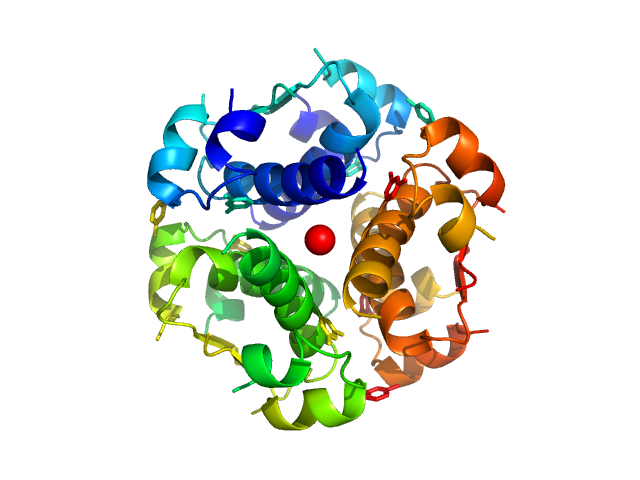
SAXS data from solutions of Human insulin hexamer from in-house SEC-SAXS in 7 mM sodium phosphate, 60 mM phenol, 200 µM Zn(CH3COO)2, 23 mM NaCl, pH 7.4 were collected on a Xenocs BioXolver L instrument at the Copenhagen University, Department of Drug Design and Pharmacology (Copenhagen, Denmark) using a 20Hz Pilatus 300K detector at a sample-detector distance of 0.7 m and at a wavelength of λ = 0.134 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
The HI sample (500 microlitres at 3.5 mg/ml) was injected onto a GE Healthcare S200 Increase 10/300 (24 ml) column at a flow rate of 0.1 ml/min at 23°C. A total of 120 x 30 second SAXS data frames were recorded throughout the HI elution. After background solvent corrections, SEC-SAXS data frames 71-83, corresponding to hexameric HI, were scaled, averaged and binned logarithmically to produce the data displayed in this entry. Additional files, including plots of the selected frames are included in the additional files of the full entry zip archive.
|
|
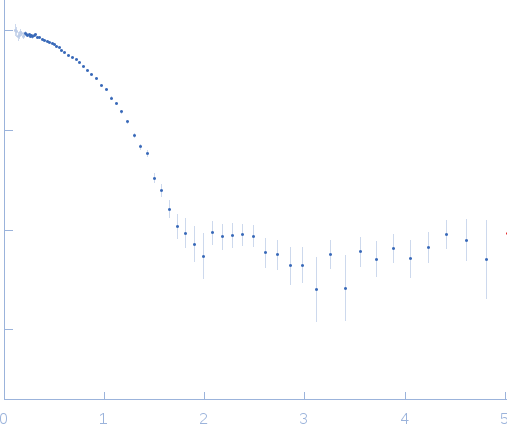 s, nm-1
s, nm-1