|
Synchrotron SAXS data from solutions of poly(aspartic acid) hydrolase-1 in 20 mM Tris pH 7.0, 100 mM NaCl, 1 mM DTT, were collected on the 12.3.1 (SIBYLS) beam line at the Advanced Light Source (ALS; Berkeley, CA, USA) using a Pilatus3 X 2M detector at a sample-detector distance of 2.1 m and at a wavelength of λ = 0.1127 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 55.00 μl sample at 8 mg/ml was injected at a 0.60 ml/min flow rate onto a Shodex KW-800 series column at 20°C. Eight successive 3 second frames were collected through the sample-elution peak. The data were normalized to the intensity of the transmitted beam and radially averaged and the the scattering of an appropriate solvent-blank was subtracted.
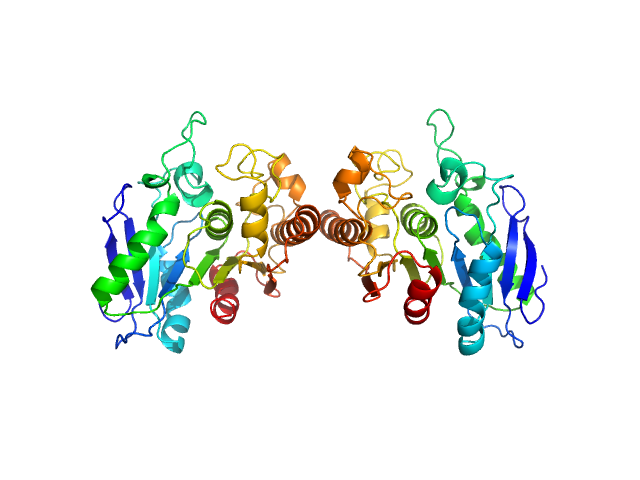
This data collection was part of a SEC-SAXS-MALLS run, where the MALLS data provided an experimental mass of 60.9 kDa. The dimer model was obtained directly from the asymmetric unit of Protein Data Bank entry 6VE6. The corresponding fit to the SAXS data was calculated using FoXS.
|
|
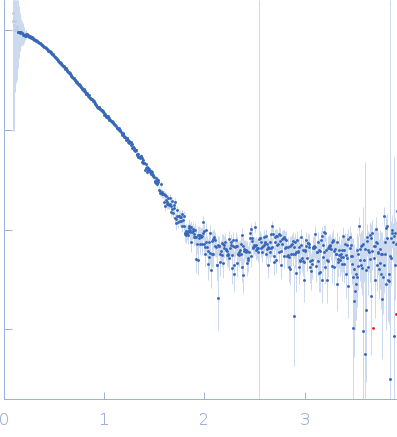 s, nm-1
s, nm-1