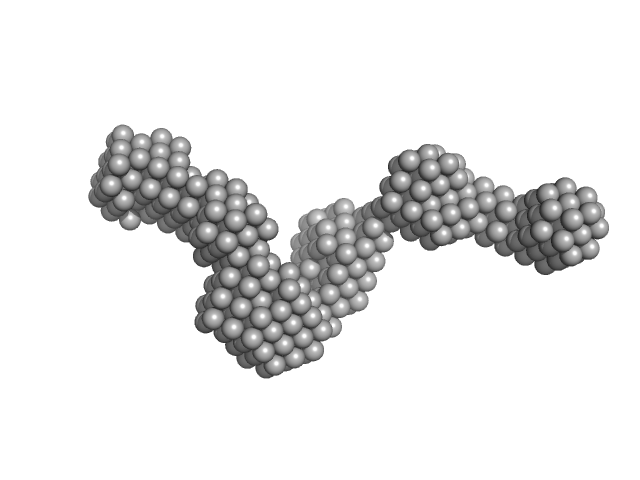
|
Synchrotron SAXS data from solutions of GH18 chitinase (ChiA) in 50 mM Tris, 250 mM NaCl, 0.25 mM DTT, pH 8 were collected on the BL4-2 beam line at the Stanford Synchrotron Radiation Lightsource (SSRL; Menlo Park, CA, USA) using a Pilatus3 X 1M detector at a sample-detector distance of 1.8 m and at a wavelength of λ = 0.1127 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). One solute concentration of 5.00 mg/ml was measured. 10 successive 1 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
Experimental temperature: UNKNOWN.
|
|
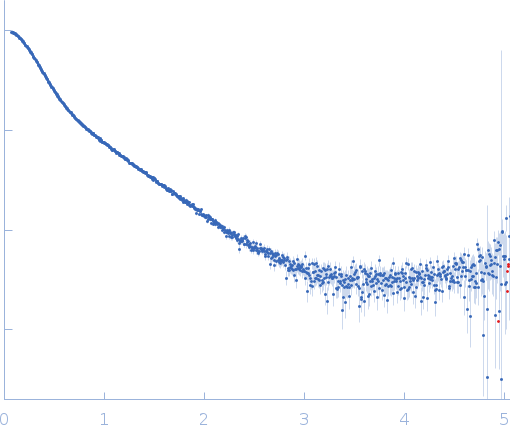 s, nm-1
s, nm-1