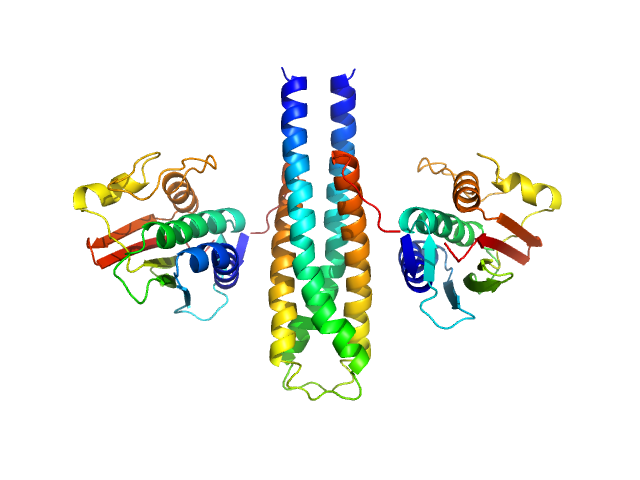
|
SAXS
data from solutions of
Sporulation kinase A
in
50mM Tris, 200mM NaCl, 150mM Imidazole, pH 8.5
were collected
on the
Bruker Nanostar II instrument (Australian Nuclear Science and Technology Organisation/Australian Centre for Neutron Scattering, Sydney, Australia)
using a Bruker HI-STAR detector
at a sample-detector distance of 65 m and
at a wavelength of λ = 0.15418 nm
(I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle).
One solute concentration of 3.70 mg/ml was measured
at 20°C.
Three successive
3600 second frames were collected.
The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
X-ray scattering data from the from the autokinase domain of the histidine kinase KinA in 50 mM Tris-HCl 200 mM NaCl 150 mM imidazole pH 8.5 were collected on a Bruker Nanostar instrument at the Bragg Institute (Australian Nuclear Science and Technology Organisation, Lucas Heights, Australia) using a HiStar 2D detector (I(s) vs s, where s = 4π sin θ/λ and 2θ is the scattering angle; λ=0.15406 nm). Approximately 15 µL of a 3.7 mg/ml protein solution was loaded into a quartz capillary mounted in a stainless steel holder. Three sucessive 3600s second frames were collected, and the buffer was collected in an analogous fashion. The data were radially averaged, and the scattering of the solvent-blank was subtracted. The data are on an arbitrary scale, and the mass of the protein was determined using a lysozyme secondary standard at a concentration of 17.5 mg/ml. The model and corresponding fits include are derived from a rigid body model using SASREF.
|
|
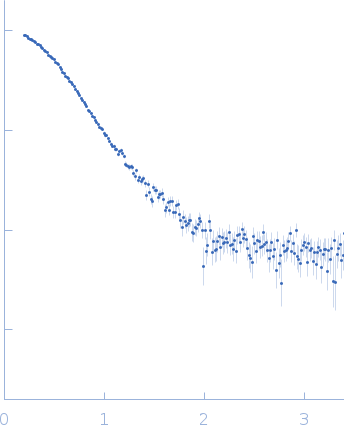 s, nm-1
s, nm-1