|
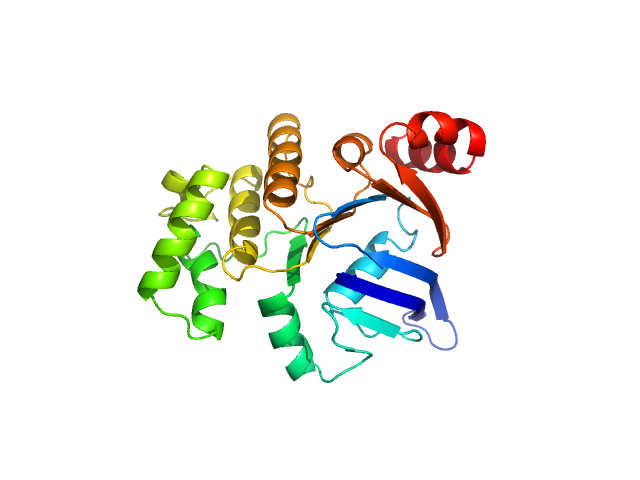
SAXS data from solutions of SaNsrF in 100 mM HEPES, 150 mM NaCl, 10% glycerol, pH 8 were collected using a Xenocs Xeuss 2.0 Q-Zoom instrument equipped with a Pilatus3 R 300 K detector and a GENIX 3D CU Ultra Low Divergence X-ray beam delivery system (Center for Structural Studies, Heinrich-Heine-University, Düsseldorf, Germany). A sample-detector distance of 0.55 m was used and a X-ray wavelength of λ = 0.154 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). Solute concentrations ranging between 0.5 and 4.2 mg/ml were measured at 15°C. 12 successive 600 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted. The low angle data collected at lower concentration were merged with the highest concentration high angle data to yield the final composite scattering curve.
Samples were injected in the Low Noise Flow Cell (Xenocs) via autosampler. For each sample, twelve frames with an exposure time of ten minutes were collected. By comparing these frames, we excluded the possibility of aggregation and radiation damage during the measurement. Data were scaled to
absolute intensity against water.
|
|
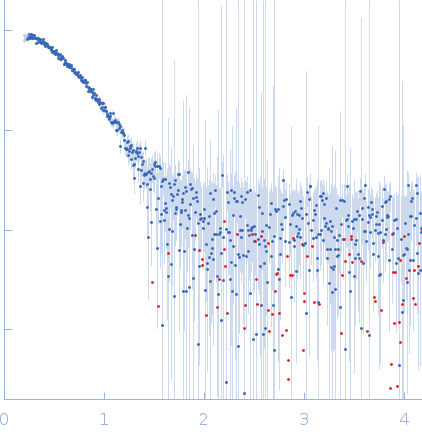 s, nm-1
s, nm-1