|
SAXS data were collected in the bioSAXS beamline B21, at Diamond Light Source, Harwell, United Kingdom. For the SYT1C2AB, 45 μl of sample with a concentration of 10 mg/ml, were injected onto a Superdex 200 increase 3.2/300 column at 20°C using 50 mM Tris, pH8, 50 mM NaCl as the running buffer. The output flow from the Agilent HPLC was directed through a 1.6 mm diameter quartz capillary cell held in vacuum. The flow rate was set to 0.08 mL min-1 and 620 frames (with an exposure time of 3 s) were collected using a PILATUS 2M (Dectris, Switzerland) detector at the distance of 4.014 m from the sample. The collected two-dimensional images were corrected for variations in beam current, normalized for exposure time and processed into one- dimensional scattering curves using GDA and the DAWN software (Diamond Light Source, UK). The background was manually subtracted using the program ScÅtter and CHROMIXS (ATSAS).
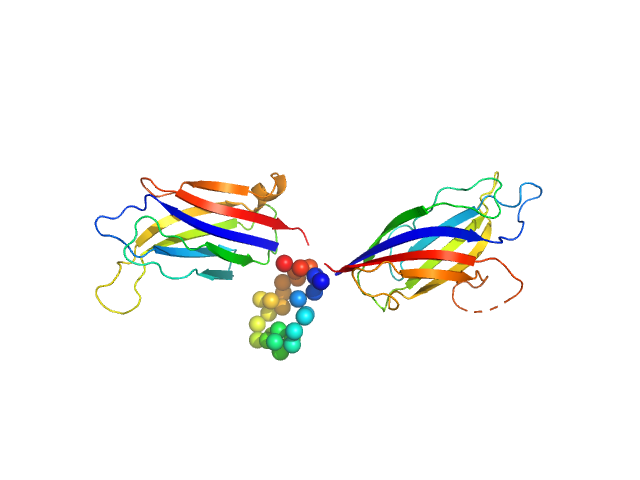
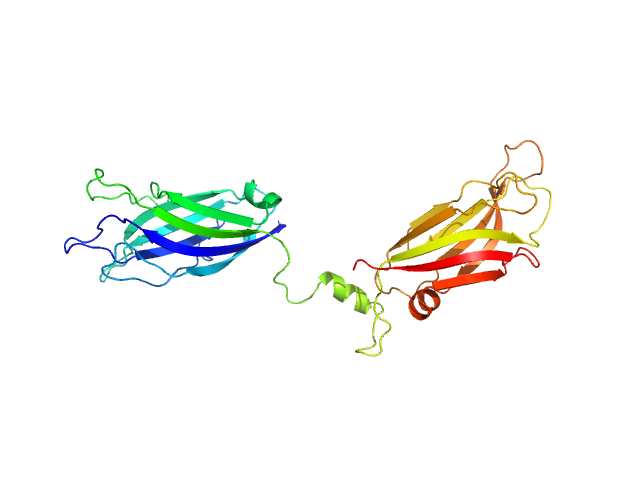
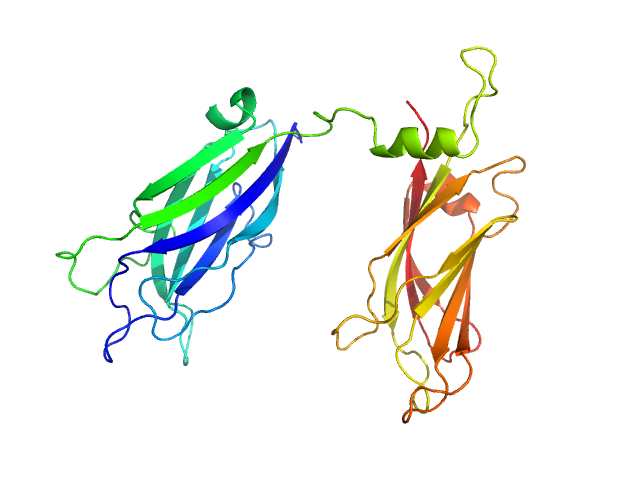
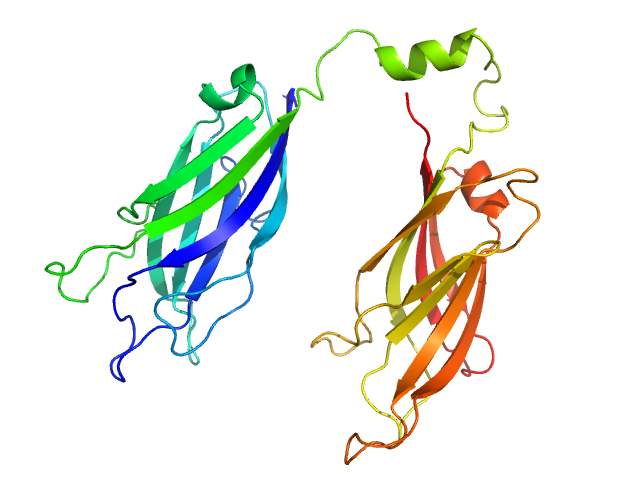
To build the low-resolution model of the C2AB fragment, we first used CORAL considering C2A and C2B as two independent domains joined by a flexible linker of variable length. However, the agreement of the resulting models with the X-ray scattering curves were not satisfactory. Hence, we carried out a modeling step to select among a set of random conformations those whose average scattering intensity best approximates the experimental SAXS data by using EOM. The resulting model includes two independent conformations that together fit to the experimental data. The analysis of the superposition of the resulting low-resolution models reveals an additional hinge point at the linker between C2A and C2B that contrasts with the crystallographic data on E-Syt2, in which the C2A and C2B domains are arranged as a rigid tandem displaying a “V-shaped structure” that is not altered by the presence of the SMP domain or Ca2+.
|
|
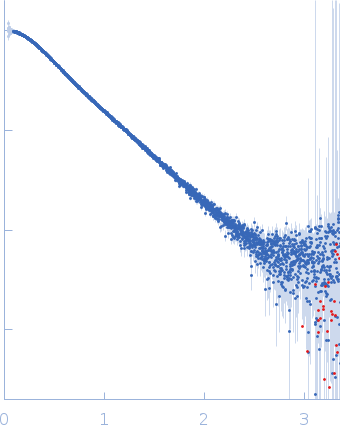 s, nm-1
s, nm-1