|
SAXS was performed at BioCAT (beamline 18ID at the Advanced Photon Source, Chicago) with in-line size exclusion chromatography (SEC-SAXS) to separate sample from aggregates and other contaminants thus ensuring optimal sample quality. The protein sample was loaded onto a Superdex 200 Increase 10/300 GL column, which was run at 0.7 ml/min by an AKTA Pure FPLC (GE Healthcare Life Sciences) and the eluate after it passed through the UV monitor was flown through the SAXS flow cell. The flow cell consists of a 1.0 mm ID quartz capillary with ~20 µm walls. A coflowing buffer sheath is used to separate sample from the capillary walls, helping prevent radiation damage (62). Scattering intensity was recorded using a Pilatus3 X 1M (Dectris) detector which was placed 3.64 m from the sample giving us access to a s-range of 0.046 nm-1 to 3.6 nm-1. 0.5 s exposures were acquired every 2 seconds during elution and data was reduced to 1D profiles using BioXTAS RAW 1.6.0. Overlapping peaks within the SEC-SAXS elution profile were deconvolved into the constituent components using evolving factor analysis (EFA) with default settings as implemented in RAW. Validity of the overall deconvolution was assessed based on the mean error weighted chi^2 for the whole deconvolution range and the deconvolved component concentration profiles.
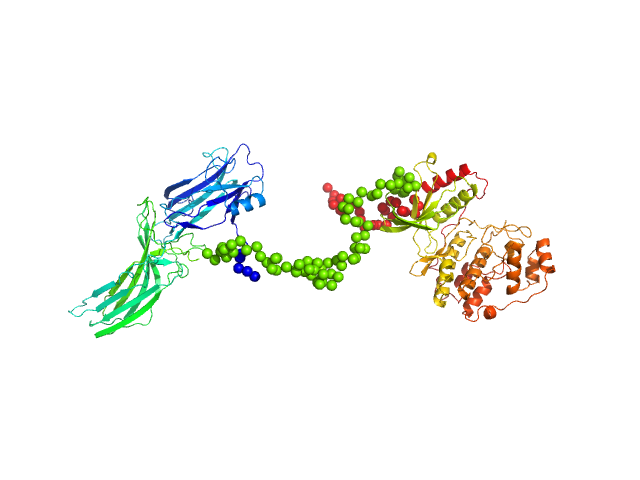
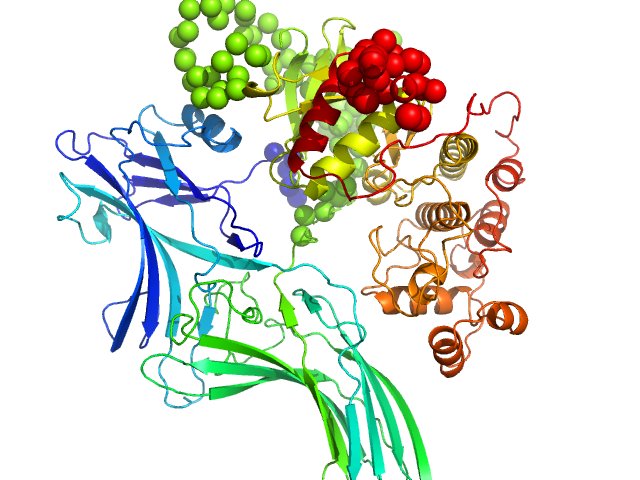
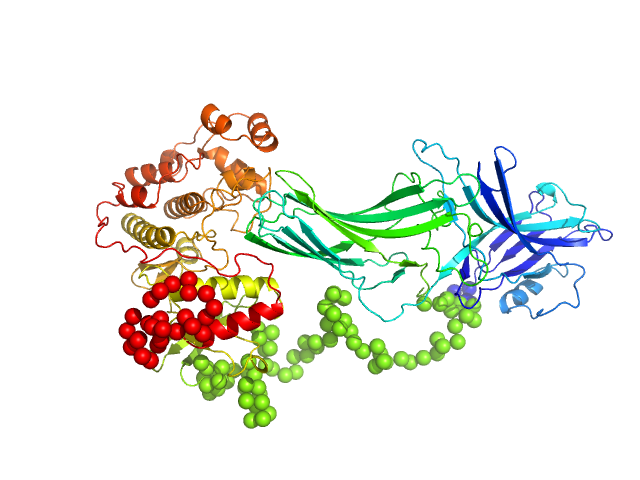
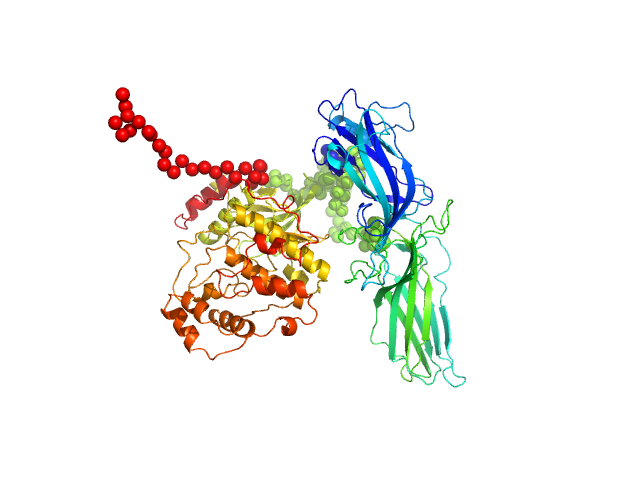
Arrestin-3 mediates desensitization and internalization of G protein-coupled receptors while the extracellular signal-regulated kinase 2 (ERK2) is a serine/threonine kinase that functions in cell growth, survival, and differentiation. It is a member of the Raf/MEK1/ERK kinase cascade. In brief, the fusion construct was cloned into a pTrc vector as follows: bovine arrestin-2 or -3 cDNA, linker with TEV cleavage site (SGLEVLFQGPGS), rat ERK2 cDNA, thrombin cleavage site (GSLVPRGSGS), and 10X His-tag. For arrestin-3: UniProt Accession #: P32120-2 (ARRB2 – BOVIN); Amino acid range 1-393; UniProt name: Beta-arrestin-2; Bovine arrestin-3 isoform short; Monomer MW: 44.17 kDa. For ERK2: UniProt Accession #: P63086-1 (MK01_RAT); Amino acid range 1-358; UniProt name Mitogen-activated protein kinase 1; Mapk1/ERK2; Monomer MW 41.28 kDa (known to dimerise).
|
|
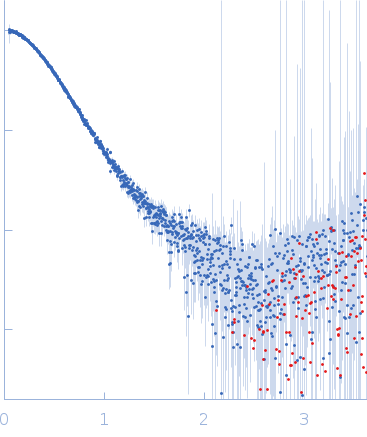 s, nm-1
s, nm-1
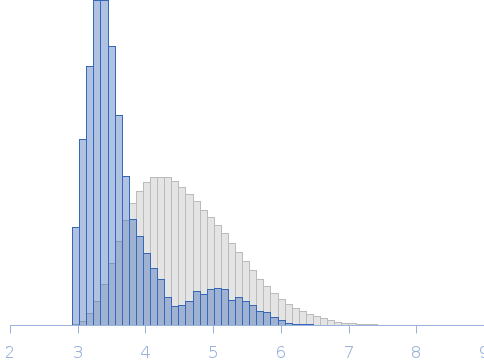 Rg, nm
Rg, nm