|
Synchrotron SAXS data from solutions of mycocerosic acid synthase in its apo form and at neutral pH in 50 mM Tris-HCl, 50 mM NaCl, 10% glycerol, pH 8 were collected on the SWING beam line at the SOLEIL storage ring (Saint-Aubin, France) using a AVIEX PCCD170170 detector at a sample-detector distance of 2.5 m and at a wavelength of λ = 0.1033 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 40.00 μl sample at 3 mg/ml was injected at a 0.15 ml/min flow rate onto a Agilent Bio SEC-3, 300 Å column at 12°C. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
Number of frames = UNKNOWN
|
|
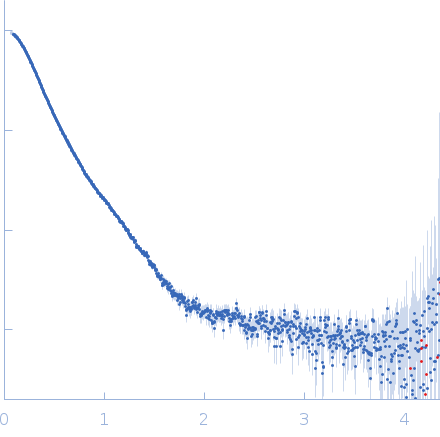 s, nm-1
s, nm-1
