|
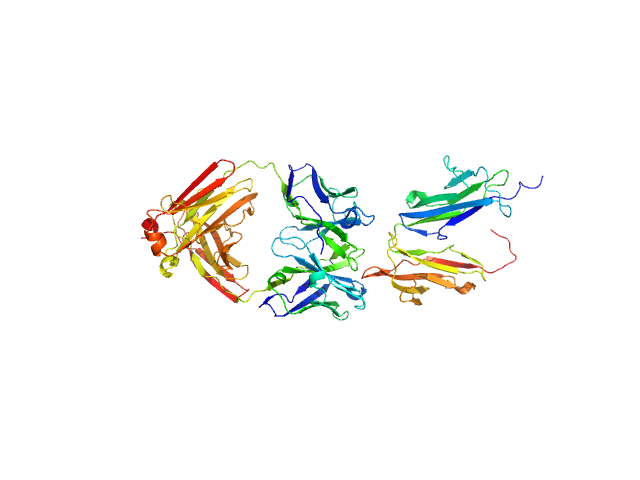
Synchrotron SAXS data from solutions of deglycosylated Fc gamma receptor IIb (CD32b) extracellular domain in complex with human IgG1 5C05 F(ab') in 50 mM HEPES, 150 mM KCl, pH 7.5 were collected on the BM29 beam line at the ESRF (Grenoble, France) using a Pilatus 1M detector at a sample-detector distance of 2.9 m and at a wavelength of λ = 0.099 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). In-line size-exclusion chromatography (SEC) SAS was employed. The SEC parameters were as follows: A 45.00 μl sample at 8 mg/ml was injected at a 0.25 ml/min flow rate onto a GE Superdex 200 10/300 column at 20°C. 600 successive 2 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
|
|
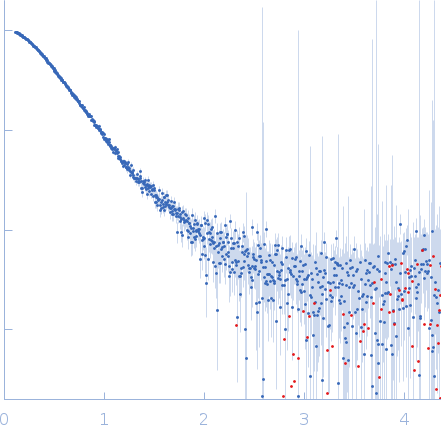 s, nm-1
s, nm-1