| MWI(0) | 321 | kDa |
| MWexpected | 308 | kDa |
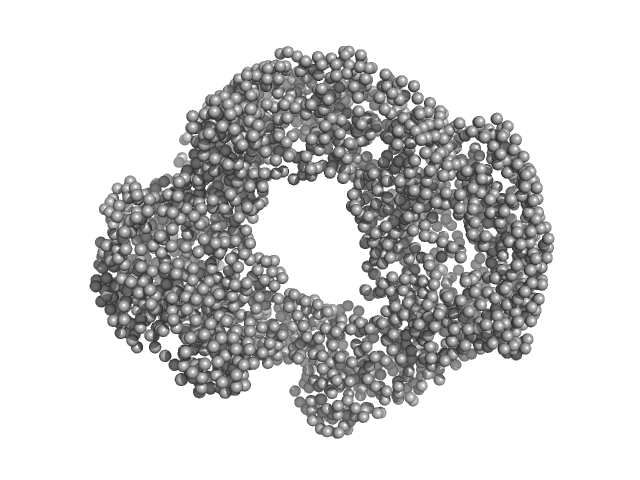
| VPorod | 424 | nm3 |
|
log I(s)
2.31×101
2.31×100
2.31×10-1
2.31×10-2
|
 s, nm-1
s, nm-1
|
|
|
|
|
|

|
|

|
|
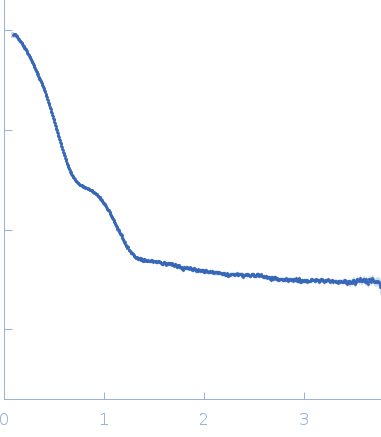
Synchrotron SAXS data from solutions of S9C peptidase from Geobacillus stearothermophilus in 10 mM Tris, 100 mM NaCl, pH 8 were collected on the BL-18 beam line at INDUS-2 (Indore, India) using a MAR 345 Image Plate detector at a sample-detector distance of 2.2 m and at a wavelength of λ = 0.10332 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). One solute concentration of 8.00 mg/ml was measured at 25°C. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
The recombinant S9Cgs protein was successfully purified using metal immobilization chromatography and size exclusion chromatography. The size exclusion chromatography revealed that the protein eluted at a volume corresponding to tetramer. Before conducting the SAXS measurements, the concentrated sample was verified to be monodisperse through dynamic light scattering. X-ray exposure time: UNKNOWN. |
|
|||||||||||||||||||||||||||