|
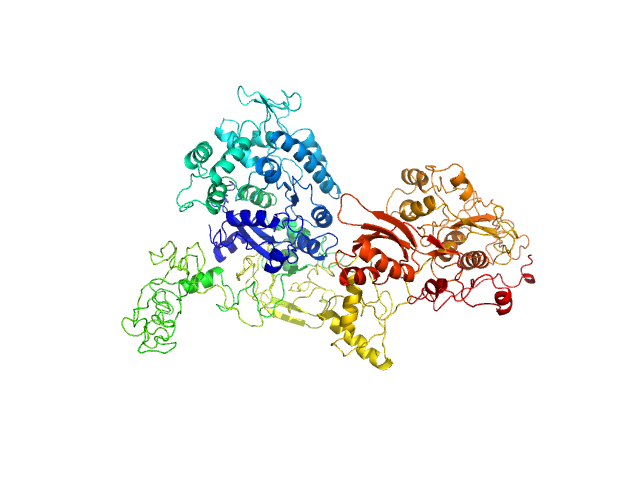
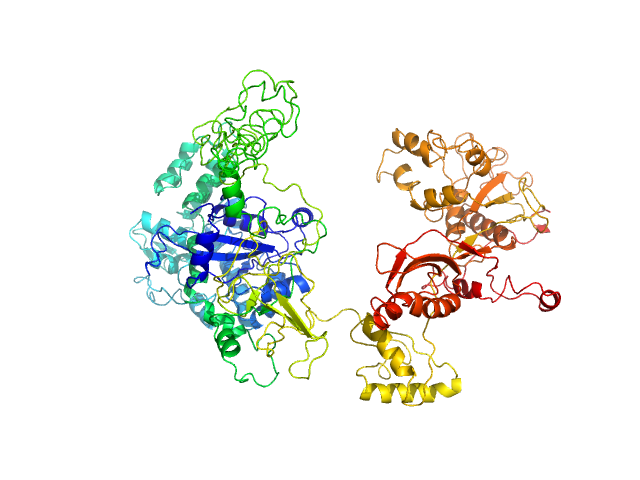
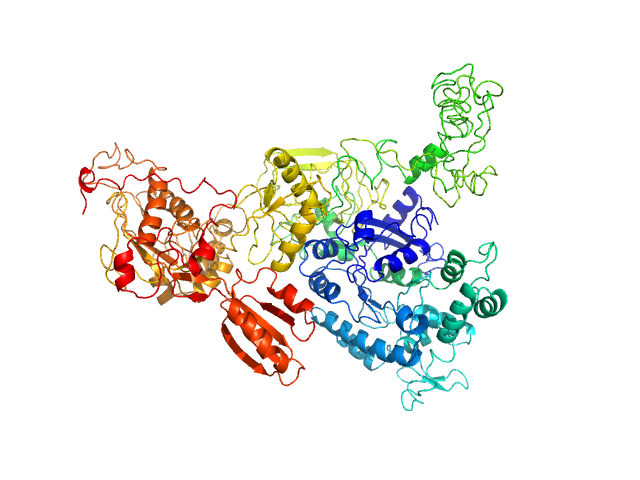
Synchrotron SAXS data from solutions of GTPase Elongation Factor like-1 protein (yeast EFL1) in 50 mM Tris pH 8.0, 10% glycerol, 300 mM NaCl and 5 mM MgCl2., pH 8 were collected using size-exclusion chromatography SAXS (SEC-SAXS) on the B21 camera at the Diamond Light Source storage ring (Oxfordshire, UK) using a Pilatus 2M detector at a sample-detector distance of 3.0 m and at a wavelength of λ = 0.124 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). The data obtained through the sample elution peak (collected as 580 successive 3 second frames) were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted and the individual subtracted data sets were scaled and averaged to generate the scattering profile displayed in this entry.
SEC-SAXS was performed at 20°C using the following parameters: Column: Shodex KW-403 ; Flow rate: 0.408 mL/min; Total acquisition time: 1740s; Sample injection concentration: 10 mg/mL; Injection volume: 45μL
|
|
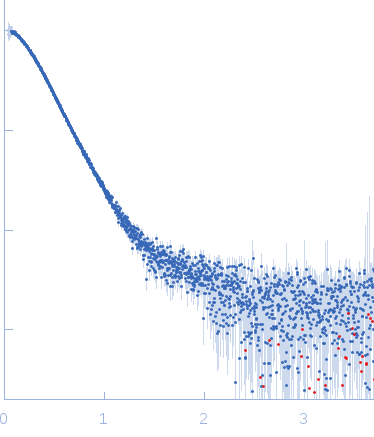 s, nm-1
s, nm-1