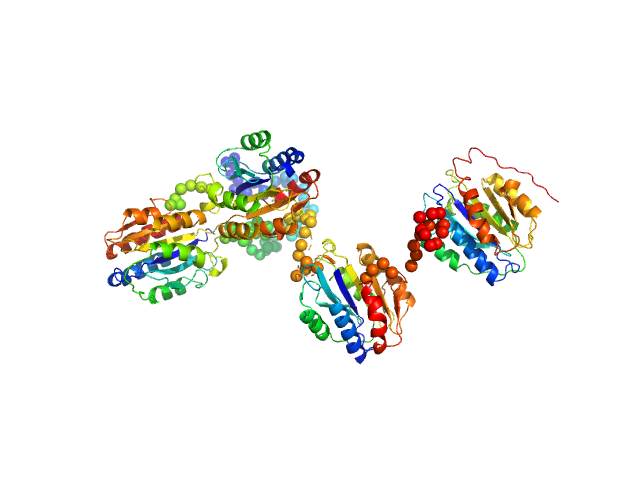|
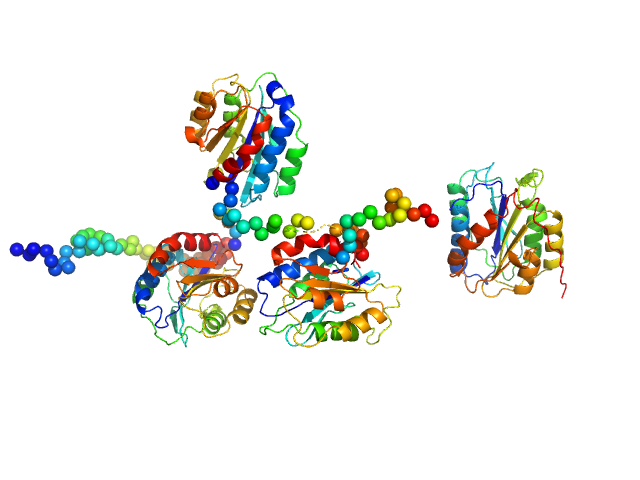
Synchrotron SAXS data from solutions of Collagen VI von Willebrand factor (VWA) four-domain fragment, N6N3 in 20 mM TRIS, 150mM NaCl 3% v/v glycerol, pH 7.4 were collected on the EMBL P12 camera at the PETRA III storage ring (Hamburg, Germany) using a Pilatus 6M detector at a sample-detector distance of 3 m and at a wavelength of λ = 0.124 nm (l(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). SEC-SAXS was performed at 20°C using the following parameters: Column: GE-Healthcare S200 Increase 10/300 analytical SEC column; Flow rate: 0.6 mL/min; Sample injection concentration: 7 mg/mL; Injection volume: 75 μL. Total X-ray acquisition time = 2400 s. The data were collected through the SEC peak of the protein as a series of 50 x 1 second exposures and processed using CHROMIXS (Panjkovich & Svergun, 2018 Bioinformatics 34(11):1944-1946) that was also used to select an appropriate matched solvent-blank. Parallel inline multi-angle laser and dynamic light scattering with refractive index measurements (SEC-MALLS/DLS/RI) were performed using a Wyatt Mini-Dawn TREOS with an in-built quasi elastic light scattering (QELS) module coupled to an OptiLab T-Rex refractometer. The quoted experiential MW for this entry is derived from MALLS. Concentration-independent MW estimates obtained directly from the SAXS data using DATMW (Hajizadeh et al., 2018 Scientific Reports 8, 7204), as part of CHROMIXS/ATSAS 2.8.3, as well as the SEC-MALLS/DLS/RI data and additional modelling files, including ITASSER homology modelling (Yang et al., 2015 Nature Methods 12, 7-8) are included in the full-entry zip archive.
SEC column = UNKNOWN. Sample injection volume = UNKNOWN. Flow rate = UNKNOWN
|
|
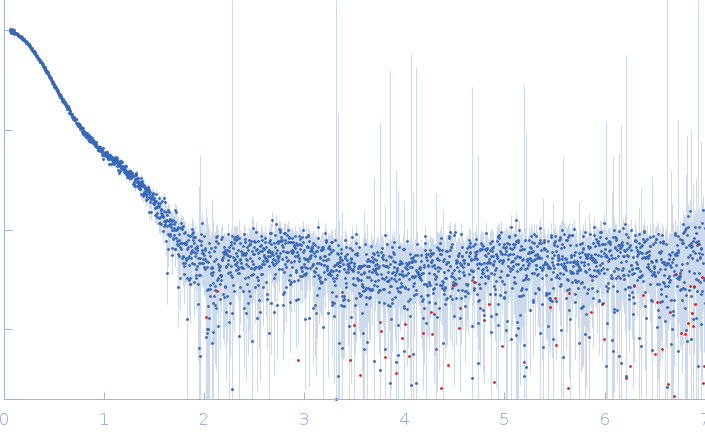 s, nm-1
s, nm-1
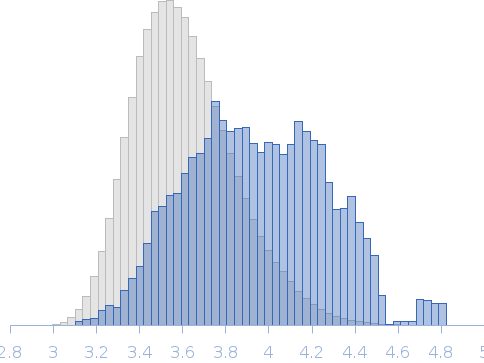 Rg, nm
Rg, nm