|
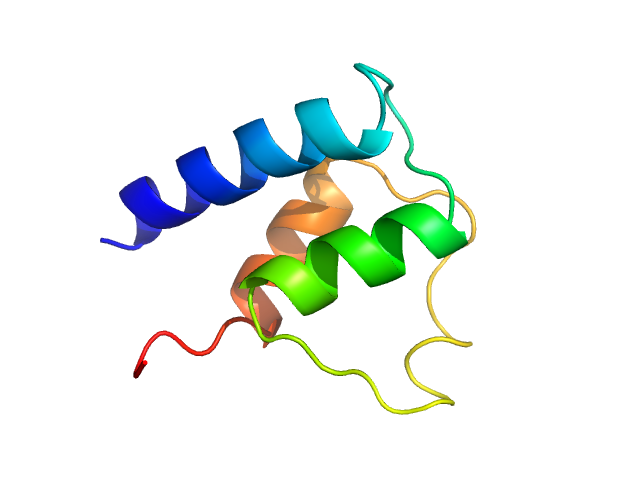
Synchrotron SAXS data from solutions of the Plasmodium falciparum myosin essential light chain N-terminal domain in 20 mM HEPES pH 7.5, 150 mM NaCl, 0.5 mM TCEP were collected on the EMBL P12 beam line at the PETRA III storage ring (Hamburg, Germany) using a Pilatus 6M detector at a sample-detector distance of 3 m and at a wavelength of λ = 0.124 nm (I(s) vs s, where s = 4πsinθ/λ, and 2θ is the scattering angle). One solute concentration of 3.75 mg/ml was measured at 20.1°C. 20 successive 0.045 second frames were collected. The data were normalized to the intensity of the transmitted beam and radially averaged; the scattering of the solvent-blank was subtracted.
|
|
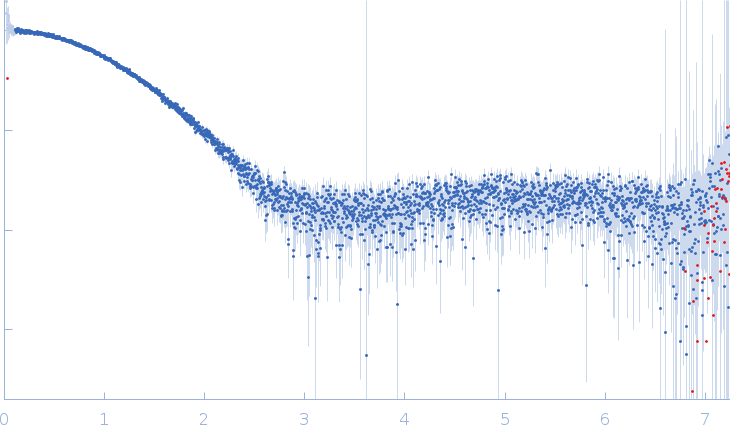 s, nm-1
s, nm-1