|
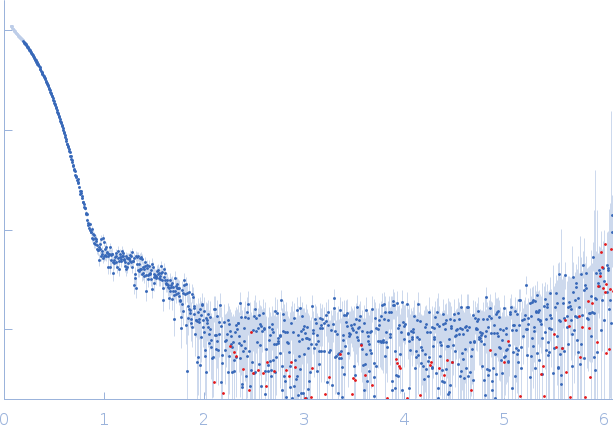
Small-angle X-ray scattering (SAXS) data collection and analysis. The SAXS data were collected on the SWING beamline36 at Soleil synchrotron (Gif-sur-Yvette, France). The TnpAWT containing fractions [50 mM HEPES (pH 7.9), 1 M NaCl, 10% glycerol, ~400mM imidazole] after elution from HisTrap column were concentrated to ~3.6 mg/ml and ~50ul of the sample was loaded onto a BioSEC3-300 (Agilent) column equilibrated in SEC buffer [50 mM HEPES (pH 7.9), 200 mM NaCl, 100 mM L-Arg HCL]. During the elution, 600 scattering measurements were taken with 1 s time frames. Data reduction, frame averaging, buffer subtraction and initial analysis were performed using FOXTROT and DATASW programs. Further interpretation of the SAXS data involved representation using the dimensionless Kratky plot and calculation of the pair-distance distribution function using the ATSAS program GNOM The excluded volume was estimated using DAMMIN in P1 and P(r) function. GASBOR program, which models the particle in solution as a protein-like assembly of dummy residues, was used to generate ab initio models with P2 symmetry constraint.
Notes - The TnpA WT protein used for SAXS includes a cMyc-His6 tag at the C-terminus.
|
|
TnpA transposase
(TnpA WT)
|
| Mol. type |
|
Protein |
| Organism |
|
Bacillus thuringiensis serovar thuringiensis str. IS5056 |
| Olig. state |
|
Dimer |
| Mon. MW |
|
116.8 kDa |
| |
| UniProt |
|
M1QZ58
(1-987)
|
| Sequence |
|
FASTA |
| |
|
 s, nm-1
s, nm-1
