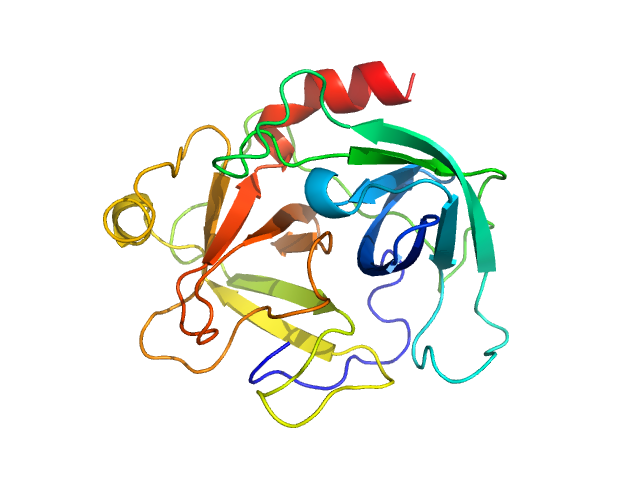
UniProt ID: P08311 (22-245) Cathepsin G
|
|
|
|
| Sample: |
Cathepsin G monomer, 25 kDa Homo sapiens protein
|
| Buffer: |
20 mM HEPES, 140 mM NaCl, pH: 7.4 |
| Experiment: |
SAXS
data collected at 12.3.1 (SIBYLS), Advanced Light Source (ALS) on 2023 Jun 3
|
S. aureus Eap is a polyvalent inhibitor of neutrophil serine proteases.
J Biol Chem 300(9):107627 (2024)
Mishra N, Gido CD, Herdendorf TJ, Hammel M, Hura GL, Fu ZQ, Geisbrecht BV
|
| RgGuinier |
1.9 |
nm |
| Dmax |
6.9 |
nm |
| VolumePorod |
27 |
nm3 |
|
|
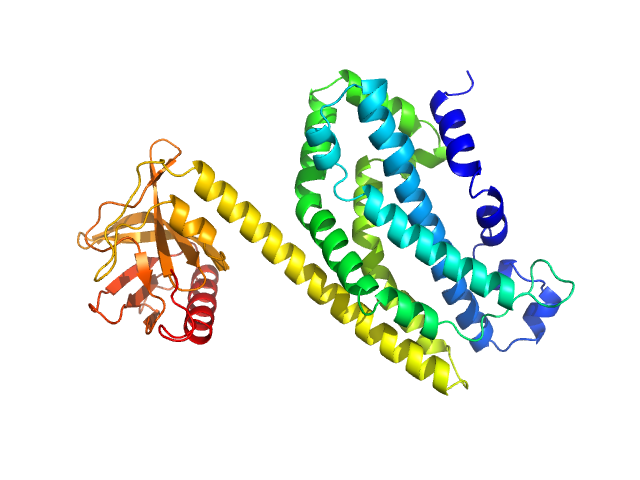
UniProt ID: Q92974-1 (190-582) Isoform 1 of Rho guanine nucleotide exchange factor 2
|
|
|
|
| Sample: |
Isoform 1 of Rho guanine nucleotide exchange factor 2 monomer, 47 kDa Homo sapiens protein
|
| Buffer: |
20mM Tris-Cl, 150mM NaCl, 1mM DTT, pH: 7.5 |
| Experiment: |
SAXS
data collected at B21, Diamond Light Source on 2021 Feb 19
|
Structural basis of microtubule-mediated signal transduction.
Cell (2025)
Choi SR, Blum TB, Giono M, Roy B, Vakonakis I, Schmid D, Oelgarth N, Ranganathan A, Gossert AD, Shivashankar GV, Zippelius A, Steinmetz MO
|
|
|
UniProt ID: P0DN75 (2-132) Pigeon iron-sulfur cluster assembly 1 homolog, mitochondrial
|
|
|

|
| Sample: |
Pigeon iron-sulfur cluster assembly 1 homolog, mitochondrial, 15 kDa Columba livia protein
|
| Buffer: |
20 mM Tris-HCl, 0.15 M NaCl, 10 mM 3-mercapto-1,2-propanediol, pH: 8 |
| Experiment: |
SAXS
data collected at BL-10C, Photon Factory (PF), High Energy Accelerator Research Organization (KEK) on 2021 Jun 8
|
A hidden property of the iron-sulfur protein in the mononuclear iron-bound state: species-dependent structural ordering induced by magnetic fields.
FEBS J (2025)
Arai S, Soga S, Hirai M, Kobayashi R, Masai H, Kimura K, Maeda K, Nagashima H
|
| RgGuinier |
2.7 |
nm |
| Dmax |
7.2 |
nm |
| VolumePorod |
39 |
nm3 |
|
|
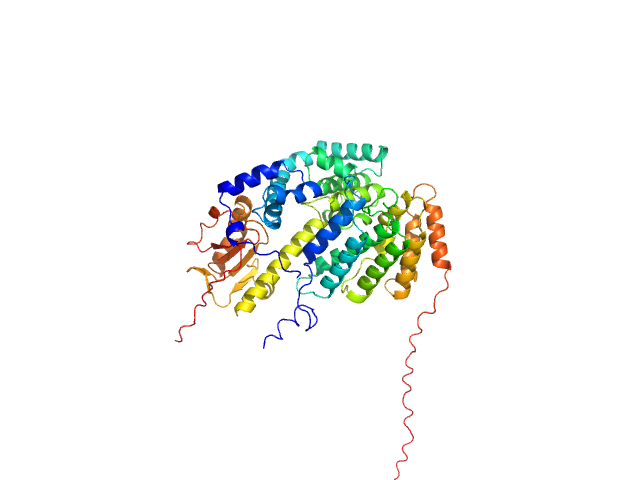
UniProt ID: Q8IWY9 (1005-1227) Codanin-1
UniProt ID: Q9Y2V0 (1-281) CDAN1-interacting nuclease 1
|
|
|
|
| Sample: |
Codanin-1 monomer, 25 kDa Homo sapiens protein
CDAN1-interacting nuclease 1 monomer, 33 kDa Homo sapiens protein
|
| Buffer: |
20 mM Tris HCl, 150 mM NaCl, 5% glycerol, pH: 8 |
| Experiment: |
SAXS
data collected at EMBL P12, PETRA III on 2023 Sep 29
|
Anemia-associated mutations disrupt the CDIN1-Codanin1 complex in inherited congenital dyserythropoietic anemia I (CDA-I) disease.
FEBS J (2026)
Stojaspal M, Brom T, Nečasová I, Janovič T, Veverka P, Verma N, Uhrík L, Hernychova L, Hofr C
|
| RgGuinier |
3.0 |
nm |
| Dmax |
10.6 |
nm |
| VolumePorod |
102 |
nm3 |
|
|
UniProt ID: Q8N4Q1 (1-45) Mitochondrial intermembrane space import and assembly protein 40
UniProt ID: O95831 (104-613) Apoptosis-inducing factor 1, mitochondrial
|
|
|
|
| Sample: |
Mitochondrial intermembrane space import and assembly protein 40 dimer, 12 kDa Homo sapiens protein
Apoptosis-inducing factor 1, mitochondrial dimer, 113 kDa Homo sapiens protein
|
| Buffer: |
25 mM HEPES, 150 mM NaCl, 2 mM TCEP, pH: 7.5 |
| Experiment: |
SAXS
data collected at 12.3.1 (SIBYLS), Advanced Light Source (ALS) on 2018 Nov 28
|
NADH-bound AIF activates the mitochondrial CHCHD4/MIA40 chaperone by a substrate-mimicry mechanism.
EMBO J (2025)
Brosey CA, Shen R, Tainer JA
|
| RgGuinier |
3.9 |
nm |
| Dmax |
12.9 |
nm |
| VolumePorod |
289 |
nm3 |
|
|
UniProt ID: Q4WF55 (1-462) N(5)-hydroxyornithine:cis-anhydromevalonyl coenzyme A-N(5)-transacylase sidF
|
|
|
|
| Sample: |
N(5)-hydroxyornithine:cis-anhydromevalonyl coenzyme A-N(5)-transacylase sidF tetramer, 223 kDa Aspergillus fumigatus (strain … protein
|
| Buffer: |
50 mM Tris, 200 mM NaCl, pH: 8 |
| Experiment: |
SAXS
data collected at Anton Paar SAXSpoint 2.0, Institute of Biotechnology, Czech Academy of Sciences/Centre of Molecular Structure on 2022 Jul 7
|
SidF, a dual substrate N5-acetyl-N5-hydroxy-L-ornithine transacetylase involved in Aspergillus fumigatus siderophore biosynthesis
Journal of Structural Biology: X 11:100119 (2025)
Poonsiri T, Stransky J, Demitri N, Haas H, Cianci M, Benini S
|
| RgGuinier |
4.0 |
nm |
| Dmax |
19.0 |
nm |
| VolumePorod |
412 |
nm3 |
|
|
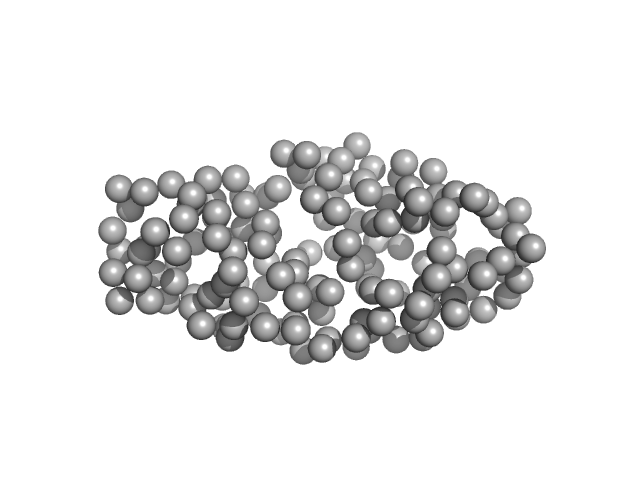
UniProt ID: Q9UBC3 (206-355) DNA (cytosine-5)-methyltransferase 3B Pro-Trp-Trp-Pro (PWWP) domain
|
|
|
|
| Sample: |
DNA (cytosine-5)-methyltransferase 3B Pro-Trp-Trp-Pro (PWWP) domain monomer, 17 kDa Homo sapiens protein
|
| Buffer: |
20 mM Tris, 300 mM NaCl, 1 mM TCEP, 5% glycerol, pH: 8 |
| Experiment: |
SAXS
data collected at 13A, Taiwan Photon Source, NSRRC on 2024 Apr 20
|
Histone modification-driven structural remodeling unleashes DNMT3B in DNA methylation.
Sci Adv 11(13):eadu8116 (2025)
Cho CC, Huang HH, Jiang BC, Yang WZ, Chen YN, Yuan HS
|
| RgGuinier |
1.8 |
nm |
| Dmax |
6.4 |
nm |
| VolumePorod |
20464 |
nm3 |
|
|
UniProt ID: P30681 (1-210) High mobility group protein B2
|
|
|
|
| Sample: |
High mobility group protein B2 monomer, 26 kDa Mus musculus protein
|
| Buffer: |
Tris 50mM, NaCl 150mM, pH: 7 |
| Experiment: |
SAXS
data collected at 13A, Taiwan Photon Source, NSRRC on 2024 Apr 27
|
High mobility group protein B2 (HMGB2)
Chun I Tu
|
| RgGuinier |
2.9 |
nm |
| Dmax |
10.1 |
nm |
| VolumePorod |
36 |
nm3 |
|
|
UniProt ID: None (None-None) Murine Immunoglobulin E (IgE) antibodies
UniProt ID: Q9STB6 (1-131) Profilin-2
|
|
|

|
| Sample: |
Murine Immunoglobulin E (IgE) antibodies monomer, 165 kDa Mus musculus protein
Profilin-2 monomer, 18 kDa Hevea brasiliensis protein
|
| Buffer: |
20 mM Tris, 50 mM NaCl, pH: 8.4 |
| Experiment: |
SAXS
data collected at B21, Diamond Light Source on 2024 Oct 3
|
Allergen-induced structural rearrangements in IgE: insights from SAXS and molecular dynamics.
Int J Biol Macromol :147658 (2025)
Gómez-Velasco H, García-Ramírez B, Siliqi D, Graewert MA, Quintero-Martinez A, Ortega E, Rodríguez-Romero A
|
| RgGuinier |
6.0 |
nm |
| Dmax |
19.9 |
nm |
| VolumePorod |
504 |
nm3 |
|
|
UniProt ID: None (None-None) β-chitin nanofibers from squid pens
UniProt ID: Q9KLD5 (24-485) GlcNAc-binding protein A (perdeuterated)
|
|
|
|
| Sample: |
Β-chitin nanofibers from squid pens None, Squid
GlcNAc-binding protein A (perdeuterated) monomer, 51 kDa Vibrio cholerae serotype … protein
|
| Buffer: |
20 mM acetate, 47% v/v D₂O, pH: 5 |
| Experiment: |
SANS
data collected at D11, ILL on 2020 Aug 18
|
Tangled Up in Fibers: How a Multidomain Lytic Polysaccharide Monooxygenase Binds Its Chitin Substrate.
ACS Appl Mater Interfaces (2026)
Sørensen HV, Montserrat-Canals M, Coder A, Prévost S, Krueger S, Vaaje-Kolstad G, Bjerregaard-Andersen K, Lund R, Krengel U
|
|
|